The Bryan Johnson Case: What Went Wrong and What We Can Learn

Bryan Johnson, the tech entrepreneur spending $2 million a year to reverse his biological age, recently shocked the longevity community by announcing a Bryan Johnson Rapamycin reset. For a man who built his entire life around the Blueprint protocol, stopping a foundational longevity drug highlights the volatile nature of extreme biohacking. This decision followed a controversial Yale longevity study 2024 results preprint that challenged the anti-aging benefits of the mTOR pathway inhibition. Johnson’s journey from “world’s most measured human” to a cautious observer offers critical insights into biological age optimization and the limits of data-driven health.
The “Reset”: Why the World’s Most Measured Human Stopped Rapamycin
Why did Bryan Johnson stop taking Rapamycin?
Bryan Johnson discontinued Rapamycin because his team concluded that the drug’s potential longevity benefits did not outweigh its significant side effects. These adverse reactions included lipid abnormalities, elevated glucose, and persistent soft-tissue infections. Additionally, a new scientific preprint suggested that Rapamycin might accelerate biological aging across several molecular markers, serving as the final justification for the pause.
The Yale Longevity Study 2024: A Critical Turning Point
The catalyst for this shift was a 2024 preprint (often associated with Yale geroscience research) that analyzed Rapamycin’s effect on 16 different epigenetic clocks in humans. This specific study claimed that Rapamycin actually accelerated aging according to these molecular markers. Dr. Matt Kaeberlein noted that the study’s conclusions were controversial because the same data claimed that proven interventions like the Mediterranean diet and exercise also “failed” to improve clock scores, raising questions about the study’s methodology.
Side Effects Observed: From Mouth Ulcers to RHR Spikes
Beyond the molecular data, Johnson reported significant physiological setbacks. This specific protocol led to Rapamycin mouth ulcers, a known side effect of mTOR inhibition that indicates the drug is reaching high systemic levels. Additionally, Johnson’s team observed resting heart rate (RHR) spikes and lipid abnormalities, suggesting that the continuous dosage was beginning to interfere with his cardiovascular and metabolic stability.

Matt Kaeberlein’s Critique: Why the Blueprint Protocol is Not Ideal
What are the primary scientific flaws in the Blueprint protocol?
Dr. Matt Kaeberlein argues that the Blueprint protocol is scientifically flawed because it attempts to test too many variables at once. This “everything and the kitchen sink” approach creates a “noise” problem where it is impossible to determine which specific supplement or habit is responsible for a biological change, making the results anecdotal rather than clinical.
The “Noise” Problem: Testing Too Many Variables at Once
The Matt Kaeberlein Blueprint critique focuses heavily on the lack of variable isolation. When a subject takes over 100 supplements daily while simultaneously undergoing intensive therapies, the resulting data is confounded. This high level of “noise” prevents researchers from identifying which interventions are providing anti-aging benefits and which might be causing long-term harm.
Biological Systems are Complicated: The Risk of Over-Inhibiting mTOR
Biological systems require a delicate balance of growth and repair signals. Chronic Rapamycin use can lead to mTORC1 over-inhibition, which starves cells of the signals needed for tissue maintenance. Furthermore, chronic dosing can inhibit mTORC2, a related pathway essential for insulin signaling, potentially leading to the very insulin resistance and metabolic dysfunction Johnson was trying to avoid.
Epigenetic Clocks vs. Functional Health: What Biohackers Get Wrong
Biohackers often treat epigenetic clock scores as the ultimate diagnostic truth. However, researchers emphasize that these clocks are research tools with a high degree of technical variance or “noise”. Functional health markers—such as VO2 max, grip strength, and immune resilience—are often more reliable indicators of vitality than a single molecular score that can fluctuate based on acute inflammation.

Analyzing the Data: When Biohacking Accelerates Biological Aging
Can extreme biohacking protocols accidentally speed up the aging process?
Yes, aggressive interventions can trigger biological stress responses that manifest as accelerated aging markers. While Johnson touted a DunedinPACE 0.64 acceleration score, suggesting he was aging at a slower rate, these metrics can be misleading if the body is simultaneously struggling with metabolic side effects or unregulated therapies that bypass traditional safety guardrails.
DunedinPACE and the Illusion of Longevity Gains
The DunedinPACE metric is a “pace of aging” tool that is highly sensitive to recent changes in health. While a score of 0.64 is impressive, experts warn that such results can provide an “illusion of longevity gains“. If the underlying system is being stressed by a massive supplement stack, the molecular clock might reflect a temporary metabolic shift rather than true, sustainable age reversal.
The “Kill Switch” Fallacy: Follistatin and Unregulated Gene Therapy Risks
One of Johnson’s most controversial moves was undergoing Follistatin gene therapy at an offshore clinic. He justified the risk by claiming the therapy had a “kill switch”. Dr. Kaeberlein dismisses this as a fallacy, noting that once exogenous DNA is introduced into human cells, a “switch” may reduce expression but cannot fully reverse potential damage if the therapy triggers cancer or a severe immune reaction.

Key Takeaways for Personalized Geroscience
How can we apply these lessons to create a safer longevity strategy?
The Bryan Johnson case teaches us that personalized geroscience must prioritize safety over speed. An ideal protocol uses intermittent dosing to allow biological recovery, tracks functional biomarkers alongside molecular clocks, and recognizes that “extreme” protocols suitable for a billionaire are often unsafe for the average person.
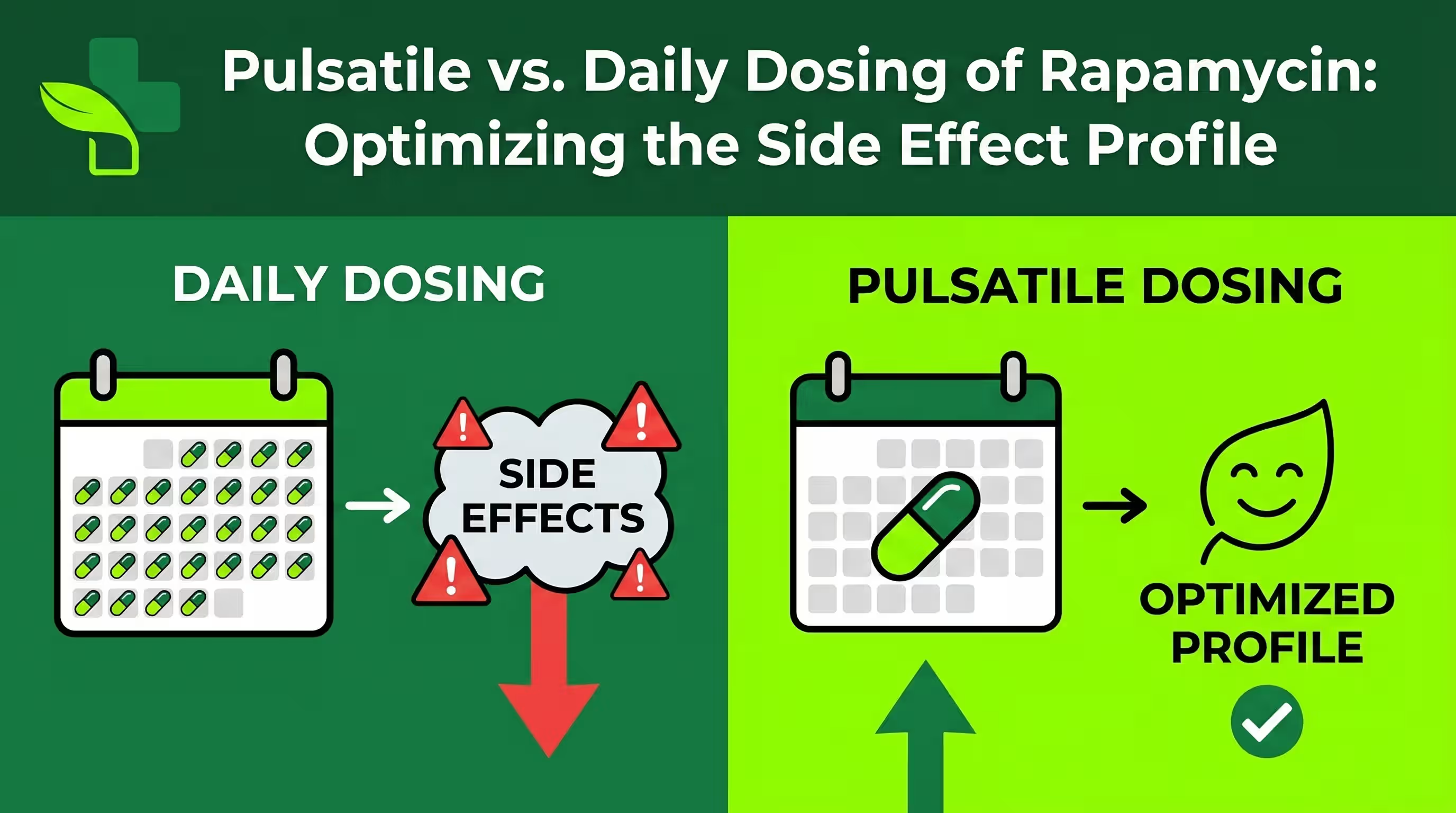
The Importance of Intermittent vs. Chronic Dosing
Most experts now favor intermittent Rapamycin dosing (e.g., once a week or cycles of several weeks on/off). This specific timing allows the mTOR pathway to reset, preventing the chronic inhibition of mTORC2 that causes metabolic side effects like insulin resistance.
Monitoring Biomarkers Beyond the Epigenetic Clock
Users should not rely solely on DNA methylation tests. It is essential to monitor standard clinical markers, including:
- Fasting Glucose and HbA1c: To detect early insulin resistance.
- Lipid Panels: To track Rapamycin-induced cholesterol changes.
- Resting Heart Rate (RHR): As a proxy for systemic stress and inflammation.
Why One-Size-Fits-All Longevity Stacks Fail
Every biological baseline is different. A protocol that works for one individual might cause a metabolic disaster for another. Personalized geroscience requires a gradual approach, introducing one variable at a time to accurately measure the body’s response before adding more complexity.
Data Presentation: Protocol and Metric Comparisons
| Feature | Chronic Dosing (Johnson’s initial path) | Intermittent Dosing (Expert Recommended) |
|---|---|---|
| mTORC1 Effect | Continuous Over-Inhibition | Periodic Suppression |
| mTORC2 Impact | High Risk of Inhibition | Minimal Impact |
| Metabolic Risk | Insulin Resistance/High Lipids | Generally Metabolic Neutral |
| Side Effect Risk | High (Ulcers, RHR spikes) | Low |
| Primary Goal | Maximum Growth Suppression | Balanced Autophagy and Repair |
Table 1: Rapamycin Dosing Strategies and Metabolic Impact

| Metric Type | Example | Strength | Weakness |
|---|---|---|---|
| Functional | VO2 Max / Grip Strength | Reflects real-world vitality | Slower to show changes |
| Molecular | DunedinPACE / Horvath Clock | Highly sensitive to change | High “noise” and variance |
| Metabolic | HbA1c / Lipid Panel | Clear clinical safety data | Indirect measure of aging |
Table 2: Functional vs. Epigenetic Metrics

The Scientific Controversy: Rapamycin and Epigenetic Markers
What does the latest research say about Rapamycin’s effect on human aging markers?
The scientific community is currently divided. While mouse models show a 13–26% increase in median lifespan across multiple studies, the 2024 Yale preprint highlighted 16 adverse epigenetic changes in humans, suggesting that the leap from mice to men is more complex than biohackers previously assumed.
What the Data Shows: 16 Adverse Epigenetic Changes
The Optispan R-Files rapamycin analysis conducted by Kaeberlein’s team scrutinized the data showing Rapamycin “aged” humans across 16 clocks. This specific finding sparked the current debate: are the clocks accurately measuring damage, or are they misinterpreting Rapamycin’s metabolic footprint as an aging signal? Until more human trials are completed, the data remains inconclusive.
Comparing Mouse Models to Human Geroscience
Rapamycin is the most successful longevity drug in animal models, consistently extending lifespan even when started late in life. However, mice in these studies are genetically identical and live in controlled environments. Humans have vast genetic diversity and variable environments, meaning a dose that is “geroprotective” for a mouse might be “toxic” for a human with a different metabolic baseline.
Conclusion: The Future of Personalized Longevity
The Bryan Johnson case is a vivid reminder that more measurement does not always mean better science. While his data-driven journey provides interesting insights, it also demonstrates the dangers of “extreme” protocols that ignore biological complexity. The future of longevity lies in personalized geroscience—using evidence-based, regulated, and thoughtfully timed interventions. By moving past the “millionaire biohacker” model, we can learn to optimize our healthspan through sustainable habits and carefully monitored therapeutics that respect the body’s need for balance.

No. Its high volume of supplements and extreme calorie restriction can cause nutrient deficiencies and metabolic stress in those without 24/7 medical supervision.
The most common are mouth ulcers (canker sores), elevated blood lipids, and temporary increases in blood glucose.
In animal models, it often reduces adiposity, but in humans, its effect on weight is variable and depends on diet and exercise.
It is a blood-based DNA methylation test that measures the “pace” at which your body is aging at the moment of the test.
Inhibiting mTOR (specifically mTORC1) triggers autophagy, the body’s cellular “cleanup” process that removes damaged proteins.
Resource links
Targeting ageing with rapamycin and its derivatives in humans: a systematic review
Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity
Epigenetic Clocks 101: Biological vs Chronological Age in Aging Research
DunedinPACE, a DNA methylation biomarker of the pace of aging
How to Track Your Biological Age Over Time
Targeting the biology of aging with mTOR inhibitors
Intermittent Administration of Rapamycin Extends the Life Span of Female C57BL/6J Mice
mTOR Signaling in Growth, Metabolism, and Disease
Evaluation of off-label rapamycin use to promote healthspan in 333 adults
Bringing Geroscience into the Mainstream: From Education to Clinical Practice, What Will It Take?