The Ethics of Longevity: Should Healthy Humans Take Rapamycin?

The transition of rapamycin (sirolimus) from a soil-dwelling byproduct of Easter Island to the center of the longevity drug ethical debate represents a paradigm shift in preventive medicine. While this macrolide remains the only pharmacological intervention proven to extend lifespan across nearly all model organisms, its increasing off-label adoption by healthy “biohackers” has ignited a fierce conflict between the cautious medical establishment and those seeking immediate geroprotection. As we navigate the biohacking rapamycin risk vs reward profile, the question is no longer just “does it work,” but whether it is ethical to prescribe a potent immunosuppressant to the healthy to delay the biological clock.
Understanding the Mechanism: mTOR Pathway and the Cellular Growth Imbalance
How does the mTOR signaling pathway contribute to biological aging and the development of chronic disease?
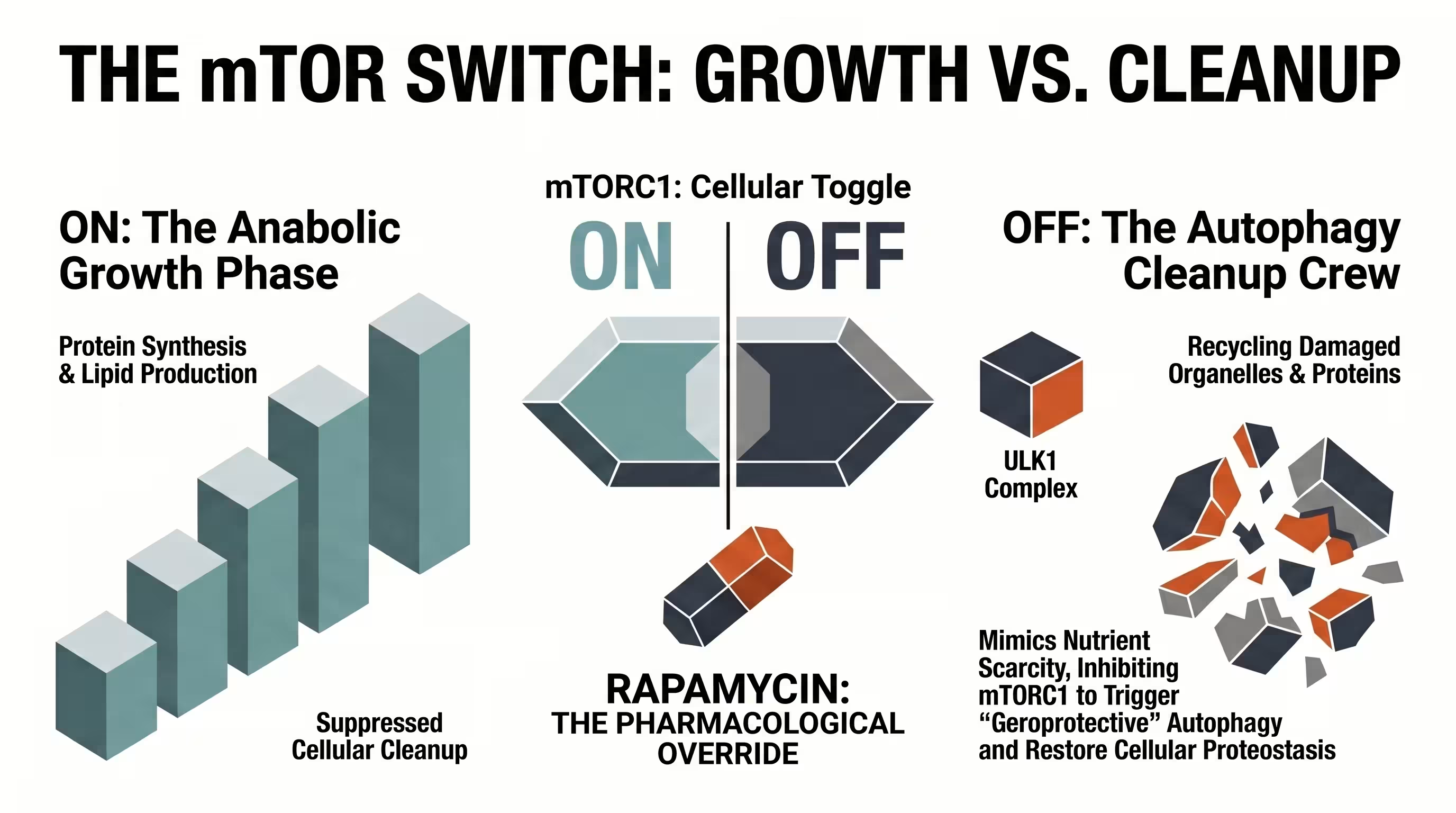
The mechanistic target of rapamycin (mTOR) acts as a master regulator that coordinates cellular metabolism with nutrient and energy availability. Aging is increasingly characterized by an mTOR-driven cellular growth imbalance, where chronically hyperactive anabolic signaling in adulthood suppresses vital “cleanup” processes like autophagy, leading to the accumulation of damaged proteins and cellular senescence.
The mTOR signaling network has evolved as a prominent regulatory hub managing crucial activities that significantly affect human lifespan. In a youthful state, the cell shifts efficiently between an anabolic phase (growth) and a catabolic phase (preservation). However, modern environments often keep this pathway “stuck” in the growth phase, a phenomenon known as antagonistic pleiotropy, where high mTOR activity is beneficial for early development but harmful in post-reproductive life.
This persistent mTOR-driven cellular growth imbalance leads to systemic decline, contributing to the development of the “four horsemen” of chronic disease: cancer, cardiovascular disease, neurodegeneration, and metabolic dysfunction. By inhibiting this pathway, rapamycin acts as a starvation mimetic, tricking the cell into prioritizing preservation and repair over proliferation. This shift triggers autophagy, a powerful molecular housekeeping mechanism that clears out dysfunctional mitochondria and misfolded proteins, thereby delaying tissue failure in the heart, liver, and immune system.
mTORC1 vs. mTORC2: Why Dosing Frequency Is an Ethical Requirement
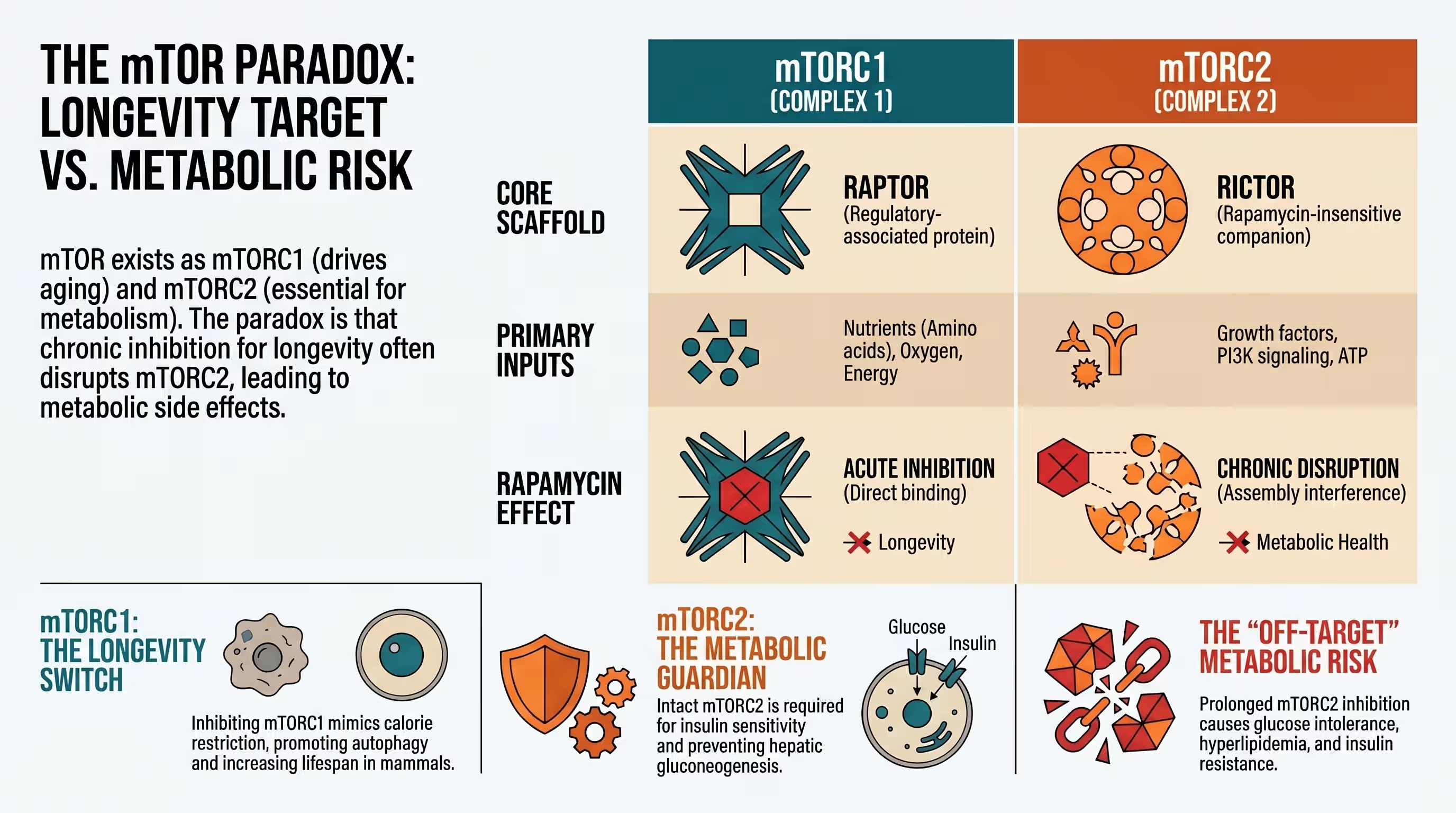
The mTOR kinase forms the core of two distinct complexes, mTORC1 and mTORC2, each with unique roles.
- mTORC1 integrates signals from amino acids, oxygen, and growth factors to drive protein synthesis.
- mTORC2 responds primarily to insulin and regulates cell survival and the actin cytoskeleton.
The ethical application of rapamycin hinges on mTORC1 recalibration—the ability to target longevity benefits while avoiding metabolic damage. The mTOR kinase exists in two distinct complexes with vastly different roles in human health.
| Feature | mTORC1 (The Longevity Target) | mTORC2 (The Off-Target Risk) |
|---|---|---|
| Primary Role | Regulates protein synthesis, lipid biogenesis, and inhibits autophagy. | Regulates cell survival, insulin signaling, and cytoskeletal organization. |
| Rapamycin Sensitivity | Highly sensitive; acutely inhibited by low, intermittent doses. | Relatively insensitive; only disrupted by high-dose or chronic exposure. |
| Inhibition Effect | Geroprotective: Promotes recycling, stem cell renewal, and genomic stability. | Metabolic Backfire: Linked to insulin resistance, hyperlipidemia, and immune dysfunction. |
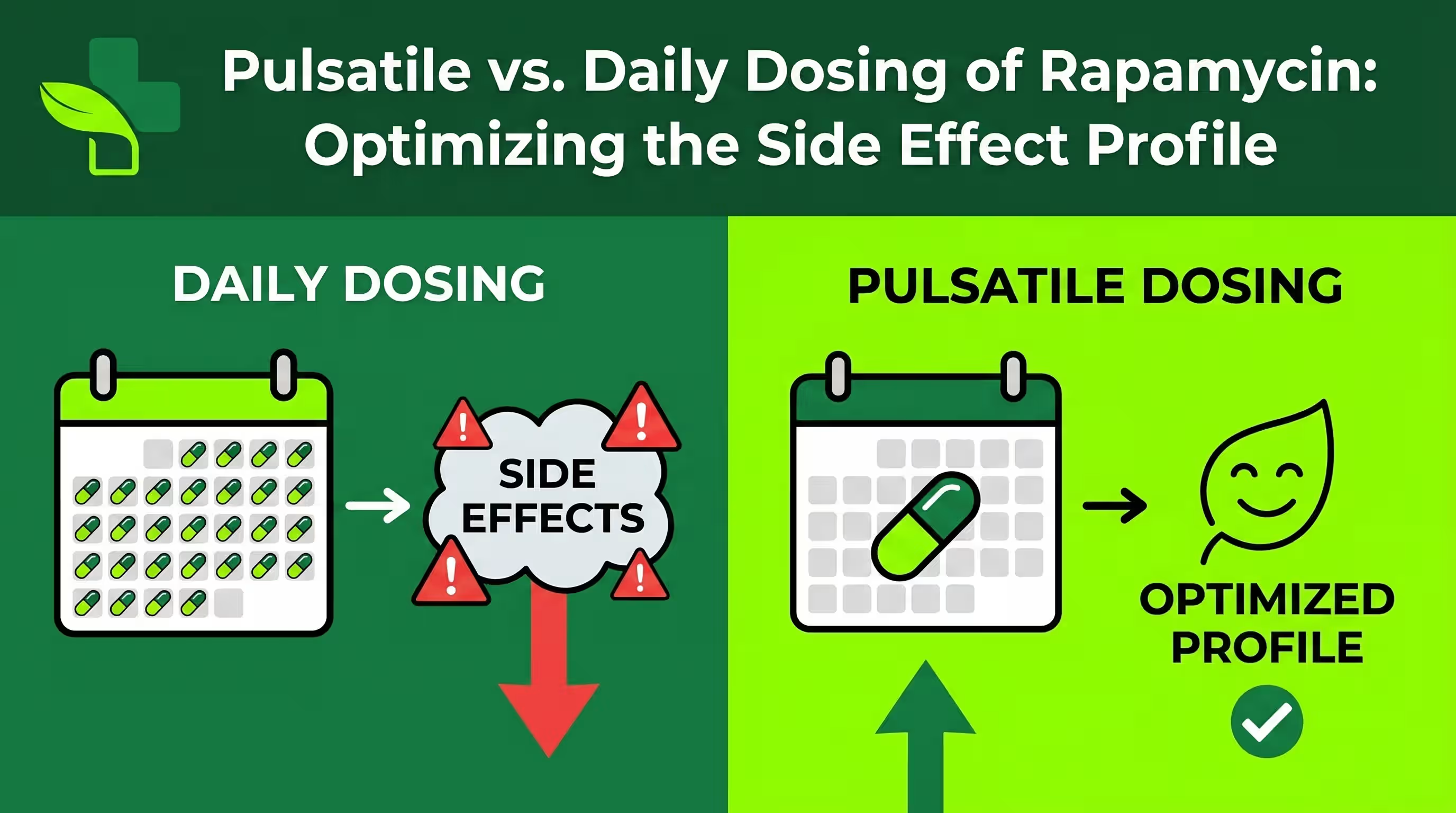
Precision dosing is an ethical necessity because RICTOR, a core subunit of mTORC2, masks the drug-binding domain, making the complex resistant to acute exposure. However, prolonged rapamycin treatment eventually prevents the assembly of new mTORC2 complexes by sequestering the available pool of free mTOR. To navigate this “trade-off problem,” ethical longevity medicine utilizes the “cycling hypothesis“, where low-dose intermittent sirolimus (typically once weekly) provides a window for mTORC2 to recover, thereby minimizing metabolic side effects like hyperglycemia while reaping the autophagy-driven benefits of periodic mTORC1 inhibition.
The Great Divide: Medical Establishment vs. Longevity Biohackers
Why is there a significant rift between mainstream medicine and the longevity community regarding the proactive use of rapamycin?
The longevity medical establishment divide stems from conflicting interpretations of evidence: biohackers argue that robust animal data justify immediate use to prevent decline, while traditional medicine remains cautious about exposing healthy populations to a potent pharmaceutical without decades of human safety trials.
Traditional “Medicine 2.0” focuses on treating symptoms after a disease is diagnosed, viewing the prescription of a powerful drug to a healthy person as an unnecessary risk. Conversely, proponents of “Medicine 3.0” argue that because aging is the primary risk factor for nearly 70% of human mortality, treating the underlying biology of aging is the only logical path to reducing human suffering.
The rise of online longevity clinics providing streamlined access to rapamycin has further widened this gap, raising concerns regarding misinformation and patient safety. The publicized case of tech entrepreneur Bryan Johnson, who discontinued rapamycin due to elevated blood glucose and susceptibility to infection, highlights the risks of bypassing peer-reviewed science in favor of anecdotal biohacking. The use of rapamycin outside established indications, especially in otherwise healthy individuals, must be accompanied with public education. These regulatory and evidentiary constraints must be considered when evaluating the future of longevity medicine.
Off-Label Risks: Immune Suppression and Metabolic Backfire
Rapamycin is a potent pharmaceutical with a significant off-label rapamycin safety profile that must be managed. It affects multiple organ systems simultaneously, and a comprehensive monitoring is highly beneficial.
- Dermatological Issues: The most common reported side effect is aphthous stomatitis (mouth ulcers).
- Metabolic Disruption: Chronic or daily use can cause a 95% rise in triglycerides and significant elevations in total cholesterol.
- Glucose Regulation: Disruption of mTORC2 impairs insulin signaling, potentially leading to a rise in HbA1c and “new-onset diabetes”.
- Immune Suppression: While low doses may boost vaccine response, excessive suppression can lower white blood cell counts and increase the risk of serious infections like pneumonia.
The Human Data Gap: Interpreting the PEARL and RAPA-EX Trial Results
Recent human trials highlight the complexity of translating animal success into healthy adult populations, showing both promise and potential drawbacks:
- The RAPA-EX-01 Trial (2026): This first randomized trial paired 6mg of weekly rapamycin with an exercise program in older adults. Surprisingly, the drug modestly blunted functional gains from training. Researchers noted a pharmacokinetic mismatch: due to rapamycin’s long ~62-hour half-life, active drug levels persisted throughout the training week, likely inhibiting the mTORC1 signaling required for muscle adaptation.
- The PEARL Trial (2025): The longest trial to date (48 weeks) found that low-dose intermittent rapamycin was generally safe in healthy adults. While it failed to significantly reduce visceral fat, women in the 10mg weekly arm saw significant improvements in lean tissue mass and self-reported pain reduction.
The Moral Argument for Geroprotection in Healthy Adults
What are the ethical justifications for healthy people taking longevity drugs to delay the biological clock?
The moral argument is built on the compression of morbidity—the goal of extending the period of life spent in vibrant health rather than chronic decline. Advocates argue it is logically inconsistent to treat the individual symptoms of aging while refusing to treat the fundamental aging mechanisms that cause them. The PEARL study provides evidence that rapamycin taken in this manner is relatively safe, and lays the foundation.
A major ethical hurdle is the “Naturalistic Fallacy“—the erroneous belief that because aging is a “natural” process, it is inherently “good” and should not be interfered with. Ethical longevity physicians argue that if we accept interventions like vaccines or heart surgery to prevent “natural” deaths, we must also accept interventions that prevent “natural” decline from cellular senescence.
Waiting for decades of definitive human data is viewed by some as an ethical failure, as it denies current generations access to potentially life-saving interventions. The goal of geroprotection is ensuring that we remain functional and independent for as long as possible, effectively adding “life to years”.
Is Aging a Disease? The Ethics of Proactive Seno-therapeutic Intervention
The debate over whether aging is a disease determines how drugs are regulated and covered by insurance. While the FDA does not currently recognize aging as a disease, the emergence of senotherapeutics—drugs that target or modulate senescent cells—provides a seno-therapeutic proof of concept for managing aging as a treatable medical condition. Rapamycin’s ability to act as a senomorphic agent positions it at the center of this proactive medical frontier.

DNA Resilience and Genomic Stability: New Findings from 2026 Research
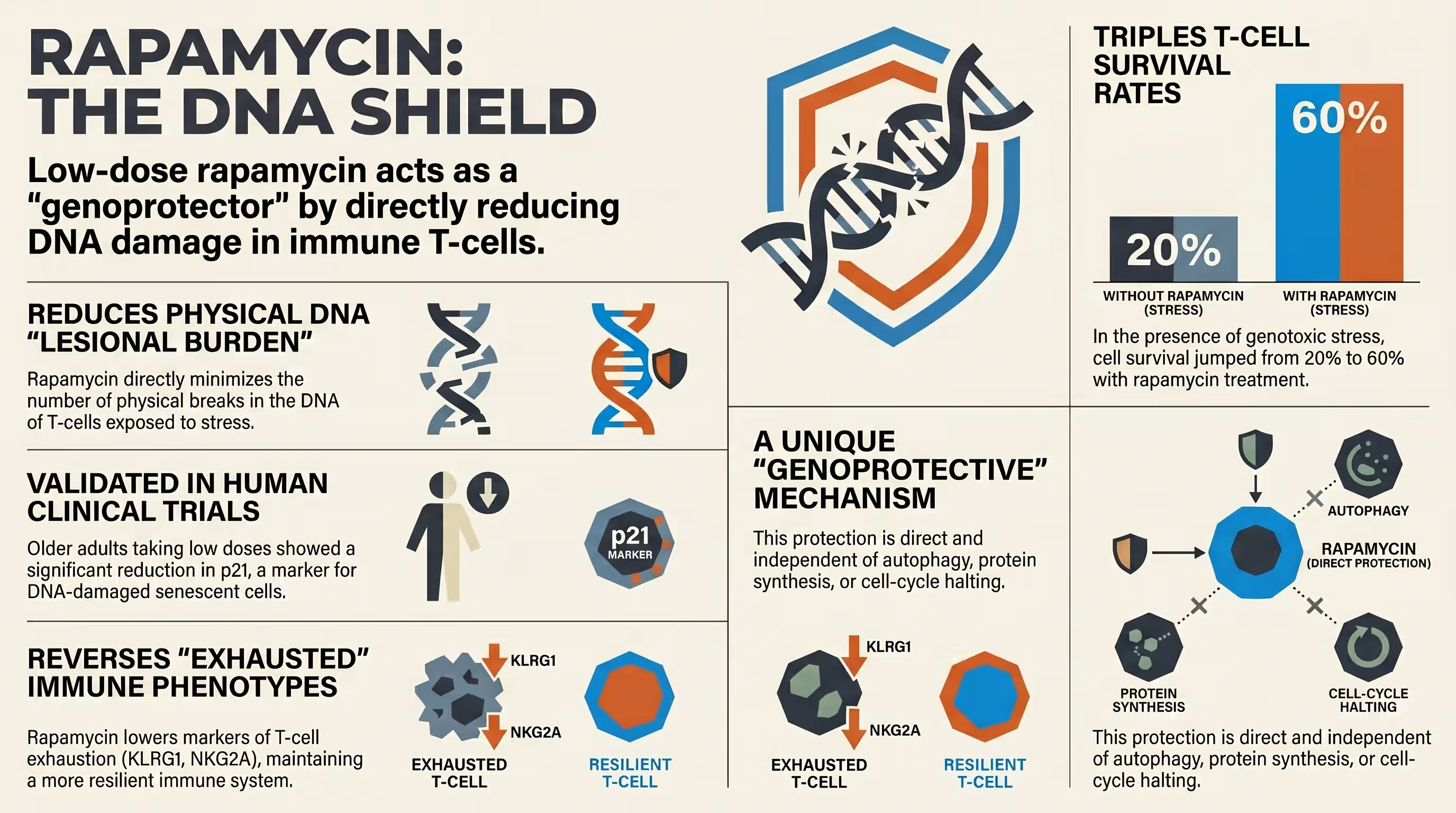
Groundbreaking research published by Kell et al. (2026) has provided a new moral imperative for proactive use. The study found that low-dose mTOR inhibition acts as a “genoprotector,” directly enhancing the resilience of human T-cells against DNA damage.
By reducing the DNA lesional burden on the immune system, rapamycin helps maintain genomic stability, a central hallmark of aging that drives systemic decline. These findings suggest that rapamycin may act as a proactive shield for the immune system before it reaches the threshold of irreversible immunosenescence. This geroprotective mechanism offers a scientific defense for using the drug in healthy adults to maintain long-term immune health.
Safety Protocols for the Ethical Longevity Physician
What are the minimum safety requirements and biomarkers for monitoring off-label rapamycin use?
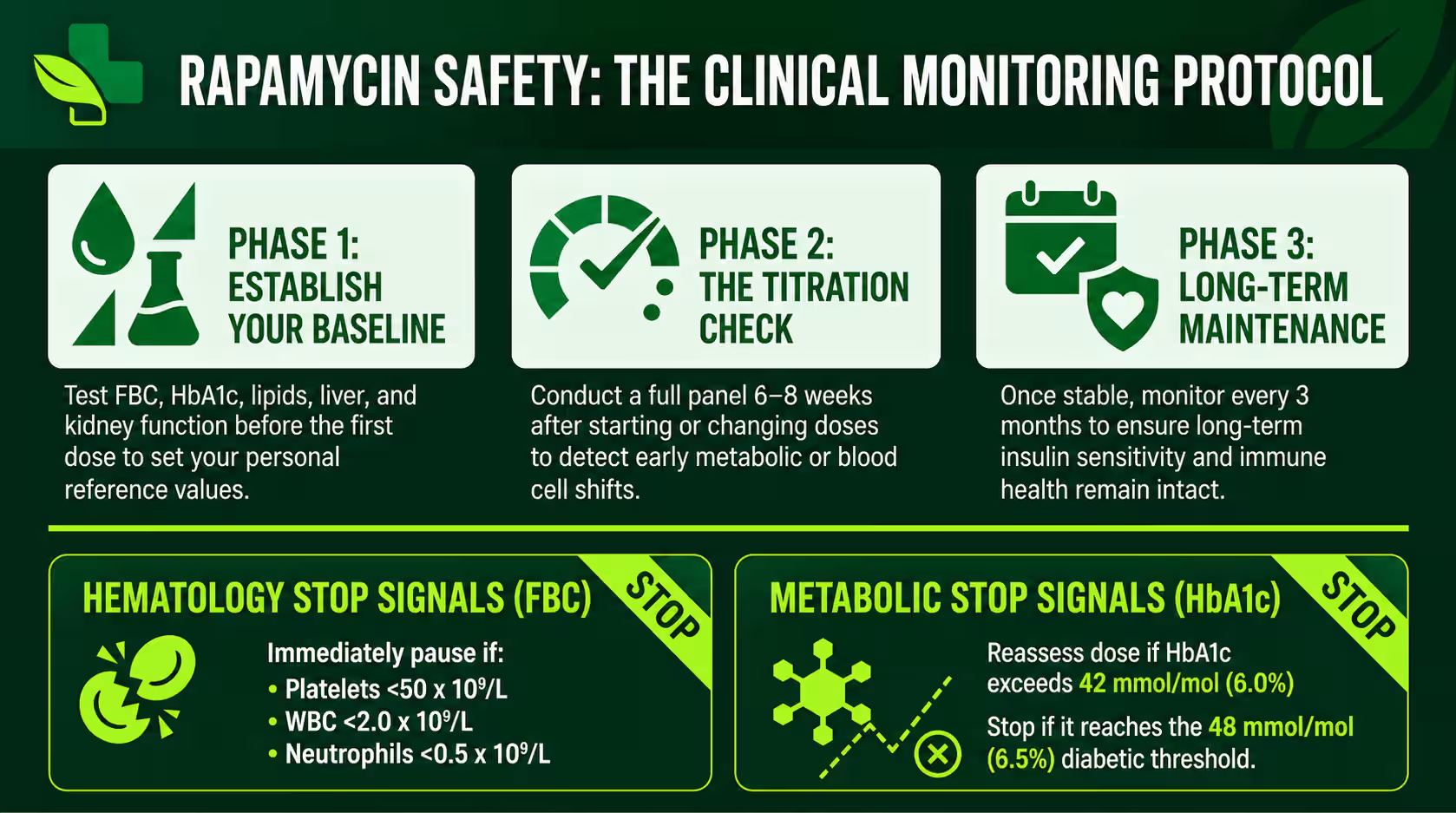
For any healthy adult choosing this path, regular blood monitoring is a safety requirement. A longevity physician must establish a safety baseline and monitor for “metabolic drift” using frequent testing to catch early safety signals before they lead to clinical disease.
Monitoring frequency must match the dosing phase. During initial titration, tests are recommended every 6–8 weeks, extending to every 3 months once stable. Because individual responses to the same dose can vary wildly, sirolimus trough levels help confirm the drug is in the intended range.
Key Biomarkers to Monitor: HbA1c, Lipid Profiles, and Hematopoietic Health
A comprehensive monitoring panel is essential to manage the drug’s potent pharmacology.
- Metabolic Regulation: HbA1c is the gold standard for detecting a drift toward insulin resistance. Any increase of more than 3-4 mmol/mol or any value above 42 mmol/mol (6.0%) warrants dose reassessment.
- Atherogenic Burden: Since rapamycin can raise triglycerides by up to 95%, monitoring ApoB and fasting lipids is critical to ensure the drug isn’t trading longevity for cardiovascular risk.
- Hematopoietic Health: A Full Blood Count (FBC) is required to monitor for dose-dependent suppression of platelets and neutrophils.
Critical “Stop Signals” for Rapamycin Use:
- Platelets: Below 100 x $10^9/L$ (thrombocytopenia risk).
- Neutrophils: Below 1.5 x $10^9/L$ (significant immunosuppression).
- Liver Enzymes: ALT/AST above 3x the upper limit of normal.
- Serious Infection: Any infection requiring antibiotics or hospitalization (e.g., pneumonia).
Final Verdict: Balancing Life Extension with Medical Prudence
The decision to take rapamycin as a healthy adult remains a calculated trade-off problem. While the potential to maintain genomic stability and delay chronic disease is compelling, the short-term risks of metabolic disruption and the potential blunting of exercise adaptations cannot be ignored. Until larger, long-term human trials are completed, the ethical path remains one of cautious interest coupled with rigorous medical oversight, consistent blood testing, and professional guidance.

It can. Chronic or high doses inhibit mTORC2, which is required for insulin signaling. In some clinical settings, this has led to “new-onset diabetes”. However, intermittent weekly dosing appears to carry a much lower risk by allowing mTORC2 to recover.
Yes. Hyperlipidemia is the most common and well-documented side effect. In transplant patients, triglycerides can rise by up to 95%. Users should monitor ApoB and may need to use concurrent statin therapy.
It is immunomodulatory. While high daily doses are used to suppress organ rejection, low intermittent doses have actually been shown to improve immune function and boost vaccine response in older adults.
Caution is required. The RAPA-EX-01 trial showed that weekly rapamycin can modestly blunt functional gains and muscle adaptation from exercise. Some users time their dose to be furthest from their most intense training sessions.
No. It is currently only FDA-approved for organ transplant recipients and specific conditions like TSC or LAM. All longevity use is considered off-label.
It is a blood test measuring the drug concentration immediately before your next dose to ensure you are staying within a safe and effective range.
Rapashop offers medical grade Rapamycin tablets, with free shipping worldwide.
Resource links
Rapamycin for longevity: the pros, the cons, and future perspectives
Influence of rapamycin on safety and healthspan metrics after one year: PEARL trial results
Targeting the biology of aging with mTOR inhibitors
Exploring the role of mTOR pathway in aging and age-related disorders
Twenty-five years of mTOR: Uncovering the link from nutrients to growth
Impact of rapamycin on longevity: updated insights
mTOR signaling in growth control and disease
Role of mTOR in Glucose and Lipid Metabolism
Targeting mTOR for Anti-Aging and Anti-Cancer Therapy
Duration of Rapamycin Treatment Has Differential Effects on Metabolism in Mice
Peter Attia MD: Disappointing results from the first rapamycin-plus-exercise trial
Should we treat aging as a disease? The consequences and dangers of miscategorisation
Senotherapeutics for metabolic disease and diabetic complications
Screening and management of metabolic complications of mTOR inhibitors in real-life settings