Rapamycin and Health Equity: Who Gets Access to Longevity Medicine?

Right now, a small group of people are already using rapamycin—off‑label and out‑of‑pocket—as a way to slow aging and improve healthspan. But behind the hype around “the first real longevity drug” lies a harder question: who actually gets to try it, and who gets left out?
This post digs into how rapamycin exposes and amplifies existing health inequities, while also exploring how pragmatic, safety‑first sourcing (including options like Rapashop.net) are starting to reshape who can realistically access long‑term rapamycin therapy.
What does “rapamycin and health equity” actually mean?
Rapamycin and health equity refers to how access to rapamycin as a longevity medicine is distributed across income, race, geography, and healthcare‑system status—not just medically, but economically and socially.
In practice:
- High‑income, tech‑savvy, insured adults in rich countries are the first adopters of rapamycin for longevity.
- Low‑income, uninsured, and racially or geographically marginalized groups are often excluded from these same options, even if they stand to benefit the most from delayed aging and chronic‑disease prevention.
This isn’t just about “who can afford a pill.” It’s about who has the knowledge, the gatekeeper‑doctors, the insurance, and the trust to even consider rapamycin as a realistic health‑span intervention.
How is rapamycin currently reaching people for longevity?
Most people today get rapamycin for longevity through one of three pathways:
- Regulated clinics and telehealth platforms that charge subscription fees, initial consultations, and include lab monitoring.
- Traditional healthcare (e.g., post‑organ‑transplant, tuberous sclerosis, or lymphangioleiomyomatosis), where the drug is prescribed but not framed as “anti‑aging.”
- Direct‑to‑consumer or research‑oriented suppliers (like Rapashop), often relying on international or generic formulations, typically without formal prescriptions.
Each of these routes has different costs, risks, and gatekeepers, which immediately creates an access gradient.
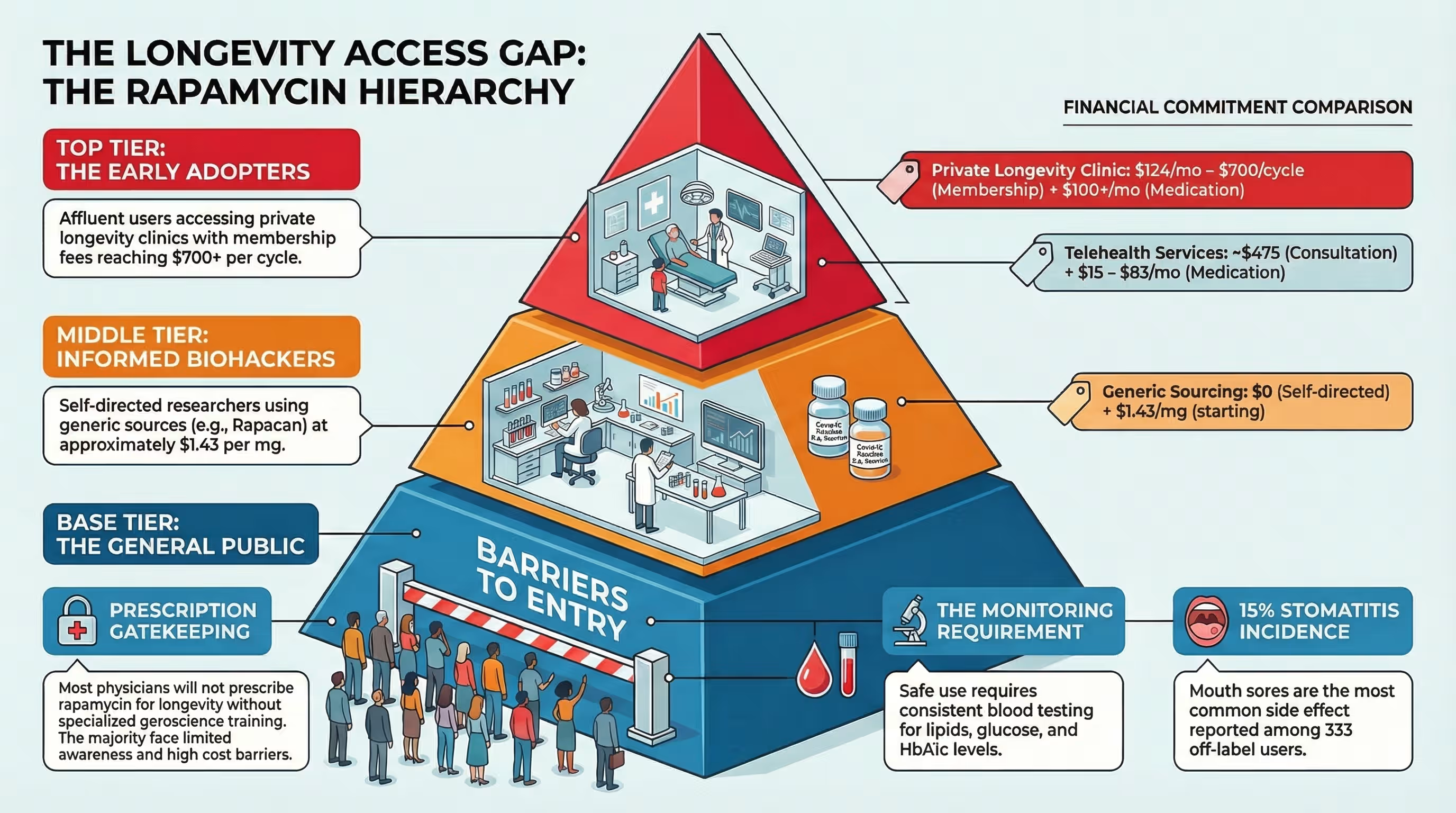
Who is actually using rapamycin for longevity today?
Based on recent surveys of off‑label users, the typical profile skews toward:
- Adults in their 40s–60s, often with higher education and above‑average income.pmc.ncbi.nlm.nih+1
- People already engaged in biohacking, longevity communities, or “preventive” health‑optimization circles.
A 2024 survey of 333 adults using rapamycin off‑label for “healthspan” showed that most dose around 3–10 mg per week, usually once‑weekly, and many report doing so without a transplant‑indication. That same cohort is also the group that can afford private labs, follow‑up visits, and the time to research protocols in detail.
Why does rapamycin create a health‑equity problem?
Rapamycin Becomes a “Two‑Tiered” Longevity Drug
Rapamycin is not a simple supplement; it’s a powerful prescription‑only immunomodulator with real side‑effect and interaction risks. Yet for longevity use, it sits in a gray zone:ema.europa+1
- For transplant patients, it’s covered when medically necessary.
- For healthy adults seeking longevity, it’s usually off‑label, self‑paid, and often funneled through expensive clinics or niche suppliers.aging-us+1
This gap between “standard care” and “longevity‑premium care” is where the equity problem crystallizes.
Cost and clinic‑based access
Several online longevity clinics such as Ageless RX now offer rapamycin for around $65–$100+ per month, sometimes higher when bundled with membership fees, labs, and “protocol” services. These models can be valuable for safety and monitoring, but they also create a financial barrier:
- Lower‑income individuals may be forced to choose between essential medications, food, or rent versus paying for a longevity‑focused rapamycin protocol.sciencedirect+1
- Clinic‑only access reinforces the idea that “real” rapamycin must be dispensed through a U.S.‑based telehealth platform, closing the door to people who lack insurance, a stable internet setup, or the time to manage another subscription.
In that context, rapamycin starts to look less like a public‑health tool and more like a luxury health‑span upgrade.
Global and pharmacoeconomic disparities
In many middle‑ and low‑income countries, rapamycin is not only unaffordable for routine use, but also tightly reserved for transplant or rare‑disease indications. Even when generic versions exist, logistics, import regulations, and lack of prescribing expertise can keep them out of reach.thelancet+3
Meanwhile, high‑income countries increasingly experiment with low‑dose, intermittent rapamycin for “healthy aging” in clinical trials such as the PEARL study, which evaluates safety and healthspan metrics over 48 weeks in normative‑aging adults. As long as these trials are concentrated in wealthy nations and affluent populations, the data and benefits will be skewed toward those same groups.
How does Rapashop fit into the access landscape?
Site‑specific data and user reports show that Rapashop has become a go‑to source for people seeking affordable, generic rapamycin (such as Rapacan 1 mg tablets) without going through a clinic‑based subscription model.
Key characteristics that distinguish Rapashop.net in the equity conversation:
- Generic / cost‑efficient sourcing: Rapacan‑brand rapamycin tablets are typically priced per‑tablet or per‑box, rather than via monthly subscriptions or bundled “programs.”
- No prescription gatekeeping: Rapashop does not require a U.S. prescription, which lowers one barrier for people who cannot easily access a longevity‑focused prescriber.
- Global shipping: Users report international orders arriving from this vendor, including in forums where people compare sourcing options.
From a strict regulatory standpoint, bypassing local prescription requirements is not without risk, which is why safety‑minded buyers often combine Rapashop‑sourced rapamycin with independent medical oversight. But from an access‑equity perspective, this model undeniably expands the circle of people who can even consider long‑term rapamycin use.

Safety, quality control, and informed self‑governance
Any discussion of off -label vendors must center on quality and safety, not just price. Several community‑driven guides on sourcing “affordable rapamycin tablets” stress key safeguards:
- Checking for Certificates of Analysis (COA) from independent labs to verify identity, purity, and absence of contaminants.
- Inspecting packaging, batch numbers, and expiry dates, and comparing photos or descriptions with known authentic products.
- Storing tablets correctly and tracking side effects, just as one would with a clinic‑dispensed prescription.
When users combine Rapashop‑sourced rapamycin with these practices—and, ideally, with a physician who understands off‑label rapamycin use—they convert a cost‑driven purchase into a more structured, semi‑supervised experiment.
What are the real risks and trade‑offs of off‑label rapamycin?
Rapamycin Is Not a Benign Supplement
Rapamycin (sirolimus) is an FDA‑approved immunomodulator with a well‑defined risk profile, originally developed for organ‑transplant immunosuppression. At low, intermittent doses used for longevity, studies and surveys suggest a relatively favorable safety profile, but not zero risk.
Key risk and side‑effect considerations:
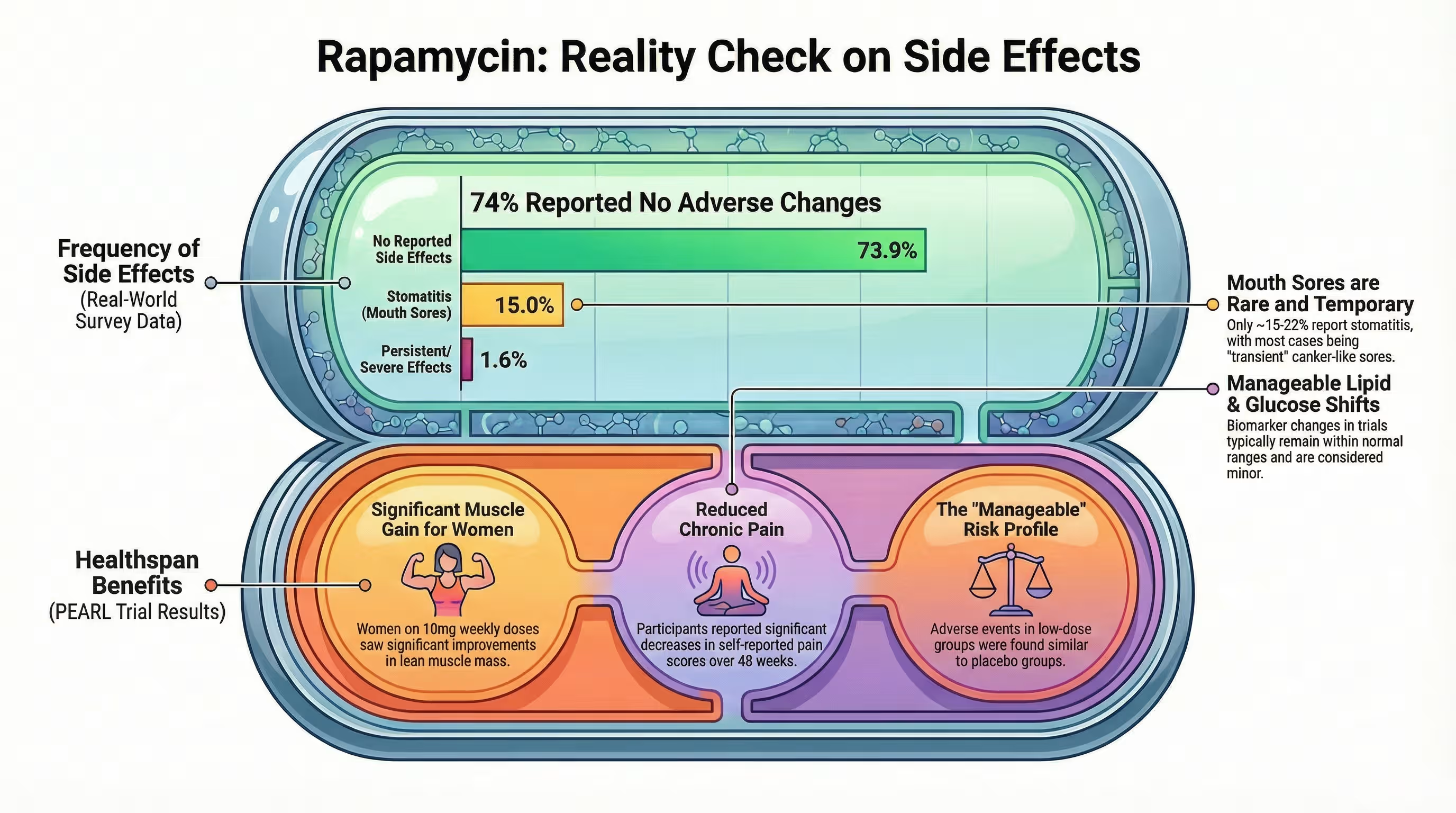
- Mouth sores (stomatitis) emerge as the most common side effect in off‑label users, reported by about 14–26% of people in recent surveys. In many cases these sores resolve or become manageable over time, but they can still be a deterrent, especially without medical or nutritional support.
- Metabolic and lipid changes such as elevated cholesterol or triglycerides can occur, though these are often mild and responsive to diet or temporary dose adjustments.
- Immunosuppression‑related concerns persist at higher or more frequent doses, including increased susceptibility to certain infections and slower wound healing, which is why clinicians generally avoid using standard transplant‑level doses in healthy adults.
Off‑label longevity use therefore involves a risk–benefit negotiation that should be individualized and, whenever possible, medically supervised.
Clinical evidence: What we know, what we don’t
Recent human data on low‑dose rapamycin for healthy adults point in a cautious but intriguing direction:
- The PEARL trial (48‑week, decentralized, placebo‑controlled) found that low‑dose, intermittent rapamycin was relatively safe in healthy, normative‑aging adults and associated with modest improvements in lean tissue mass and pain in women, though long‑term clinical benefits are still unknown.
- A 2025 review in Aging‑US concluded that clinical evidence for off‑label rapamycin in healthy adults remains limited and insufficient to broadly recommend it for longevity, underscoring the need for larger, longer trials.
In other words:
- There is early evidence of safety and some health‑span signals, but not yet definitive proof of extended lifespan or disease prevention in healthy humans.thelancet+1
- The most robust benefits are still preclinical, from mice and other animal models, where rapamycin consistently extends median and maximum lifespan.sciencedirect+1
How can rapamycin access become more equitable?
Rapamycin doesn’t have to be a luxury longevity drug by default. With deliberate policy, research, and provider choices, it can start to look more like a shared resource. Practical steps that could move the needle toward equity:
1. Inclusive clinical trials and public‑facing data
Current trials such as PEARL focus on “healthy, normative‑aging” adults who are also tech‑savvy enough to participate in decentralized, online‑heavy studies. To broaden equity:
- Trials could actively recruit diverse socioeconomic, racial, rural, and global populations, not just urban, insured, English‑speaking cohorts.
- Researchers could publish and translate plain‑language summaries of findings, so that people outside academic circles can understand benefits, risks, and realistic expectations.
2. Transparent pricing and alternative sourcing models
Equity‑aware clinicians and platforms can explicitly acknowledge that clinic‑only models exclude many people, then:
- Offer tiered pricing or sliding‑scale options for rapamycin and monitoring.
- Provide clear information about generic, off‑brand, or international options, while still emphasizing the need for quality checks and medical supervision.
This doesn’t mean encouraging prescription‑free use; it means recognizing that people will seek alternatives anyway, and equipping them with the best possible safety information.
3. Education and harm‑reduction guidance
Many people discover rapamycin through longevity podcasts, Reddit threads, and blogs, not through their primary‑care doctors. That creates a gap where misinformation can spread.
Steps to close that gap:
- Medical‑professional communities and longevity‑focused platforms can publish standardized, conservative protocols (e.g., 3–6 mg per week, weekly dosing, priority lab tests such as CBC, lipids, glucose, and renal function).
- Patient‑facing resources can explain how to identify reputable vendors, interpret COAs, and recognize red flags (e.g., prices that seem too good to be true, no contact information, or no batch testing).
4. Policy and system‑level changes
At the population level, the only way to truly equalize access to rapamycin and other longevity‑relevant therapies is through structural change:
- Universal health‑coverage systems that consider evidence‑based, off‑label uses for high‑value drugs, rather than reflexively excluding them.
- Patent‑management and generic‑drug policies that allow low‑cost rapamycin formulations to be legally available in more countries, not just as transplant drugs.
Until those systemic changes happen, the current landscape will remain tilted toward wealthier, better‑connected groups.
How can individuals navigate rapamycin access safely and ethically?
Even within an inequitable system, individual decisions still matter. If you are considering rapamycin for longevity, here are some evidence‑informed, safety‑first principles.
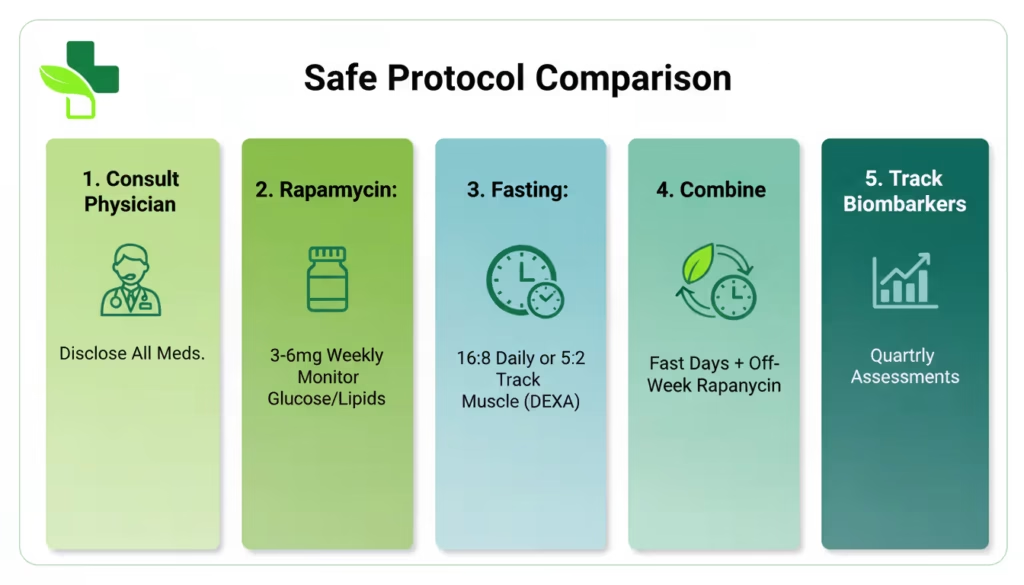
1. Start with medical consultation (when possible)
Ideally, rapamycin should be discussed with a clinician who:
- Understands its pharmacology and off‑label use for aging.
- Can review your medical history, medications, and lab baselines (e.g., kidney function, lipids, immune status).
If you live in a country or community where this kind of access is limited, online second opinions or tele‑medicine consults (even if you source tablets elsewhere) can still add a layer of protection.
2. Prioritize gradual, low‑dose regimens
Community‑collected data and surveys suggest that many people using rapamycin for longevity end up in the 3–10 mg per week range, often taken once weekly. Within that band, many clinicians and researchers lean toward the lower end (around 3–6 mg) for healthy adults, especially when starting.
A typical, cautious approach looks like:
- Starting at 1–3 mg once weekly, then titrating up over weeks while monitoring for side effects.
- Avoiding “mega‑dose” or daily‑dosing regimens unless explicitly guided by a knowledgeable clinician familiar with transplant‑level protocols.
3. Monitor for side effects and adjust accordingly
The most commonly reported side effect is stomatitis (mouth sores), followed by minor metabolic changes. Users must be aware of any viral symptoms they are experiencing too. Practical mitigation strategies are dose reduction or a temporary discontinuation. Side effects on low dose regiments are easily reversible if these basic actions are followed.

Rapamycin is a compound originally discovered in soil bacteria from Easter Island (Rapa Nui). It has been used for decades as an immunosuppressant in organ transplant patients. However, extensive animal studies have shown that rapamycin can extend lifespan and healthspan—delaying age-related decline and diseases like cancer, cardiovascular issues, and neurodegeneration. It is currently one of the most promising “geroprotective” drugs being studied for aging in humans, though it is not yet FDA-approved specifically for anti-aging.
No. The FDA has not approved rapamycin for longevity or anti-aging purposes. It is only approved as an immunosuppressant (for organ rejection) and in certain cancer therapies (e.g., for renal angiomyolipoma). However, physicians can prescribe it off-label for longevity, which is legal but controversial. This off-label use is a key driver of unequal access, as it often requires a willing doctor, disposable income, and knowledge of the research.
Access is heavily skewed toward affluent, well-educated individuals who can:
Afford off-label prescriptions (often $100–$300+ per month without insurance coverage).
Pay for concierge or longevity clinic consultations ($500–$2,000+).
Navigate online pharmacies or compounding pharmacies.
Understand the scientific literature and manage potential side effects.
Lower-income populations, rural communities, and those without strong health literacy are largely excluded, creating a significant health equity gap.
If longevity interventions become available only to the wealthy, we risk creating a “longevity divide” where rich individuals live dramatically longer, healthier lives while poor populations continue to suffer from early aging and preventable diseases. This could exacerbate existing disparities in life expectancy (e.g., the 10–15 year gap between richest and poorest US counties). Ethical longevity medicine requires equitable access—otherwise, it becomes a tool for deepening social inequality.
Cost: No insurance coverage for off-label anti-aging use.
Regulatory status: Lack of FDA approval for longevity means no standardized dosing or safety guidelines for healthy people.
Medical gatekeeping: Requires a prescription from a doctor knowledgeable and willing to prescribe off-label.
Information asymmetry: Knowledge about rapamycin circulates in biohacker, tech, and academic circles—not in primary care clinics serving underserved communities.
Risk perception: Without large-scale human trials for longevity, many doctors are hesitant to prescribe, further limiting access.
Yes, but it depends on policy and market changes. Potential pathways include:
Large-scale clinical trials proving safety and efficacy for healthy aging, potentially leading to FDA approval and insurance coverage.
Generic production (rapamycin is already off-patent) driving down costs, though manufacturing quality varies.
Public health programs or nonprofit initiatives subsidizing access.
Integration into preventive care guidelines for aging-related risk factors.
However, without proactive equity-focused policies, even cheap generics may not reach underserved populations due to lack of prescribing infrastructure and awareness.
Rapamycin is a potent drug with side effects including mouth ulcers, insulin resistance, glucose intolerance, increased infection risk, and potential immune dysregulation with long-term use. For equity-focused access, any expansion must include:
Medical monitoring (blood work, glucose checks).
Clear patient education about risks vs. potential benefits.
Screening for contraindications (e.g., active infections, diabetes).
Uncontrolled, widespread access without safeguards could harm vulnerable populations, undermining the ethical case for equity.