Is Rapamycin Effective for Long Covid or ME/CFS Symptoms? (Emerging 2025–2026 Trend)

The landscape of chronic illness treatment is shifting as researchers move beyond mere symptom management toward addressing the biological roots of exhaustion. As we move through 2026, a clear trend has emerged: rapamycin, once known primarily as a transplant drug and longevity “miracle,” is being repurposed as a precision tool for patients battling Long COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). Recent clinical data suggests that low-dose rapamycin may help resolve the cellular “traffic jams” that prevent your body from cleaning up damaged parts and restoring energy.
Does current research support using rapamycin for Long COVID or ME/CFS?
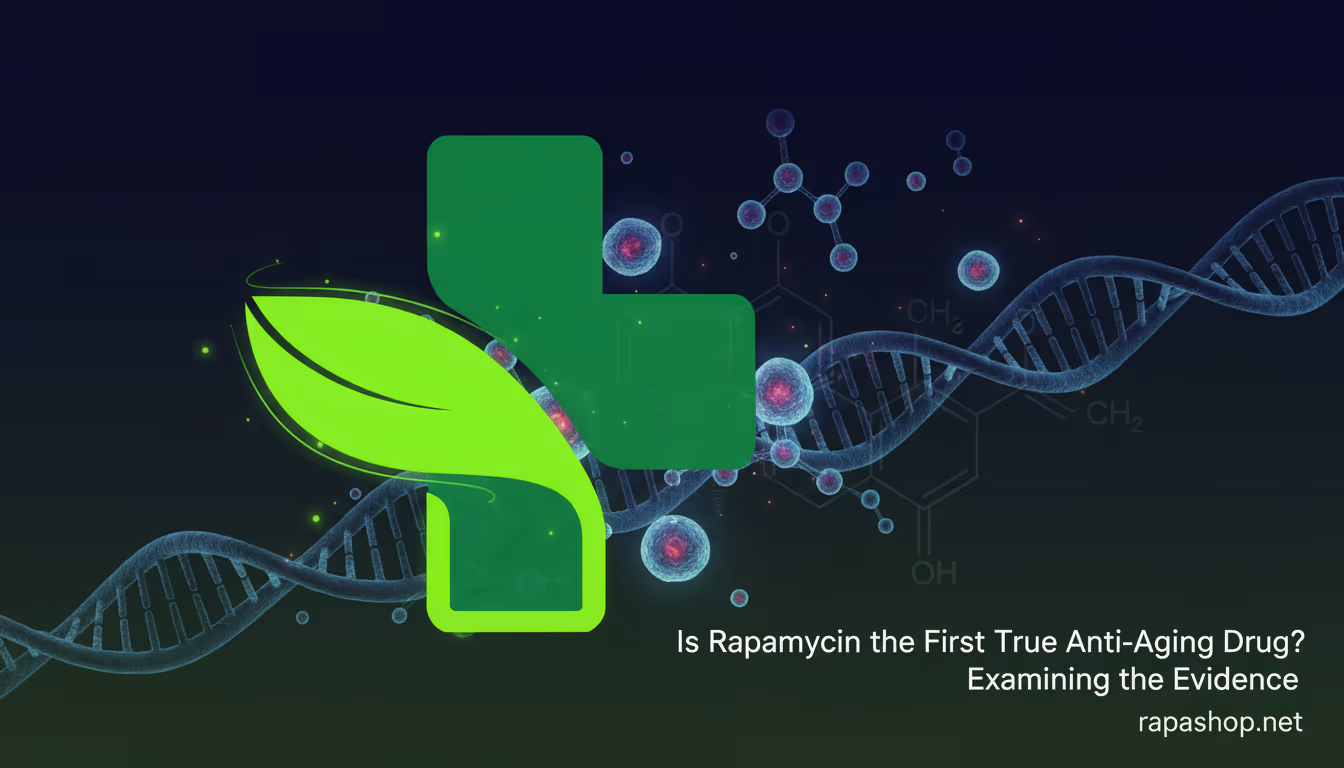
Emerging evidence from 2025 and 2026 clinical trials indicates that low-dose rapamycin can alleviate core symptoms like post-exertional malaise (PEM) and chronic fatigue by inhibiting overactive mTOR pathways. In a pivotal Simmaron Research study, 72.5% of ME/CFS patients reported significant recovery in fatigue and orthostatic intolerance. This response is highly correlated with the restoration of autophagy, the body’s cellular “waste management system”.
While these results are promising, researchers emphasize that rapamycin is not a universal cure; it appears most effective for a specific biological subgroup, particularly those whose illness followed a viral infection or those showing specific biomarkers of autophagy disruption.
How does rapamycin address the underlying biology of post-viral fatigue?
Rapamycin acts as an mTOR inhibitor, which reboots the process of autophagy to clear out dysfunctional mitochondria and viral fragments that linger in the body. In patients with Long COVID and ME/CFS, a protein called mTOR is often chronically overactivated, effectively “turning off” the cell’s ability to recycle damaged components. This leads to a buildup of cellular “garbage,” resulting in chronic inflammation and a lack of energy production.
Detailed physiological insights reveal:
- Mitochondrial Renewal: By blocking mTOR, rapamycin allows cells to initiate autophagy, replacing old, energy-depleted mitochondria with healthy ones.
- Immune Modulation: At low doses, rapamycin shifts the immune system from an “exhausted” state to an active, regulated state, potentially helping the body finally clear latent viral reactivations like EBV or SARS-CoV-2 fragments.
- Reducing Inflammation: Mechanistic studies show that rapamycin stabilizes the secretion of inflammatory cytokines, which can “cool down” a hyper-active nervous system.

What have clinical trials discovered in 2025 and 2026?
The most recent data from Simmaron Research and Mount Sinai highlights a dramatic improvement in quality of life for a subset of “responders,” particularly those with high levels of the pATG13 biomarker. In the Simmaron Phase 1 pilot, researchers observed that patients who responded to the drug showed a clear “inverse relationship” in blood markers: their levels of pSer258-ATG13 (an autophagy-blocking protein) dropped, while BECLIN-1 (an autophagy-promoting protein) rose.
| Clinical Trial | Lead Institution | Key Focus | 2026 Status |
|---|---|---|---|
| RAPA STEP 4 ME | Simmaron Research | Autophagy & PEM recovery | Phase II ongoing |
| CoRE Long COVID | Mount Sinai | Immune profiling & T-cell health | Final results expected late 2026 |
| ITP Longevity | NIA (Federal) | Survival & lifespan extension | Validated 15-20% life extension in mice |
Dr. David Kaufman, a lead clinician in the Simmaron trial, noted that about two-thirds of his patients saw “dramatic” improvements, with some previously housebound individuals returning to daily activities like walking and light exercise.
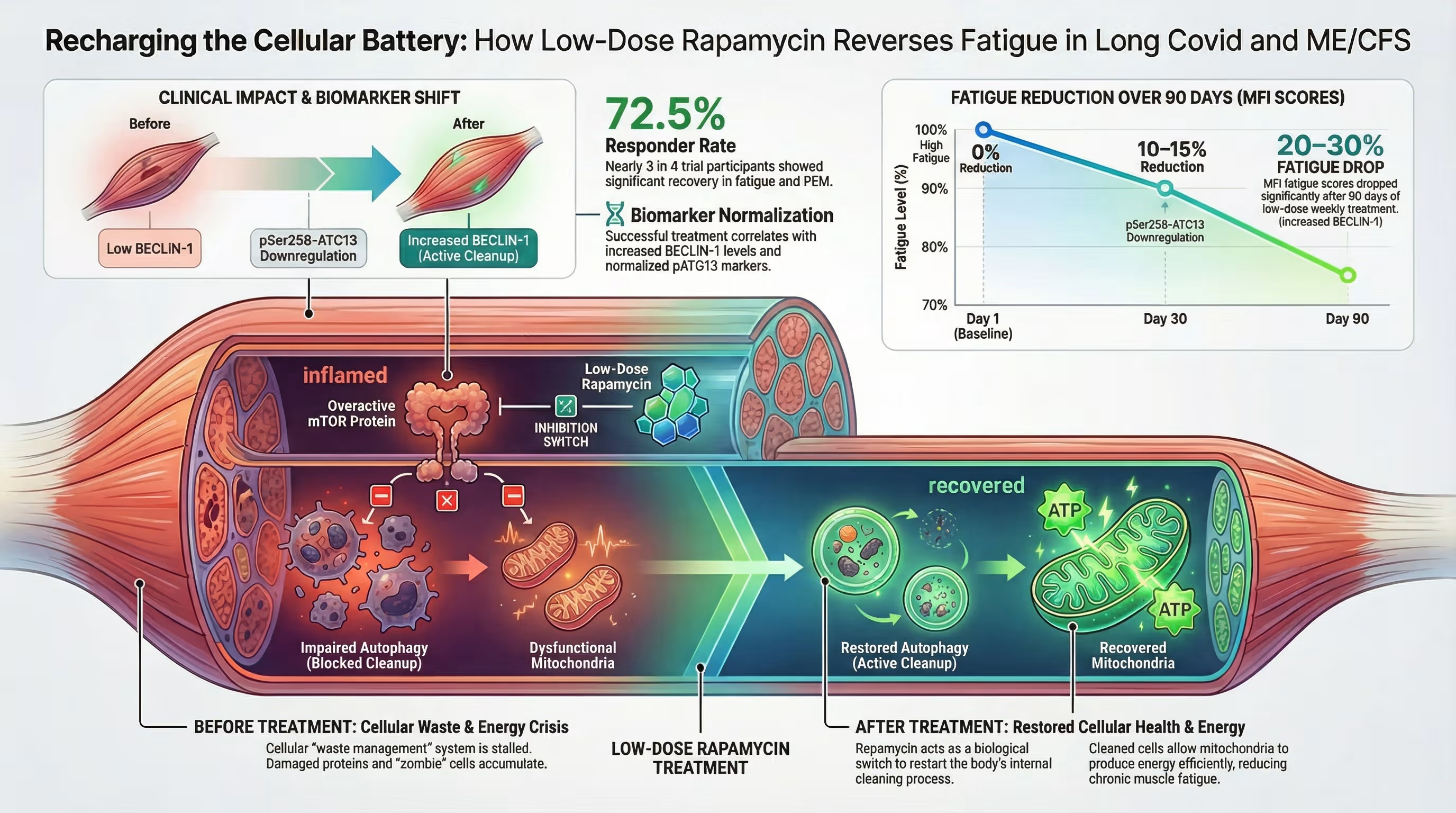
Is there a specific “Responder Phenotype” for rapamycin therapy?
Patients with a history of viral onset (such as Mono/EBV) or documented viral reactivation show the strongest positive response to rapamycin. This is a landmark discovery in 2026: we can now use biomarkers to predict who will benefit from the drug rather than relying on trial and error.
Key characteristics of a likely responder include:
- Viral Trigger: ME/CFS or Long COVID that began after a clear viral infection.
- Elevated pATG13: High levels of this specific protein in the blood indicate that the body’s autophagy “switch” is stuck in the off position.
- T-Cell Exhaustion: Dysfunctional immune cells that are “tired” from fighting a persistent or reactivated pathogen.
Real Patient Experiences: Miracles and Roadblocks
“Experience” is a critical pillar of medical trust, and user forums like Reddit provide a window into how rapamycin performs outside of controlled labs.
- The “Basketball” Success: One 26-year-old Long COVID patient, Alex, went from being bed-bound with severe PEM to playing basketball within six weeks of starting weekly 5mg doses. However, he later experienced a relapse after six months, highlighting that rapamycin may need to be part of a broader, cycled protocol.
- The “Whisper” Breakthrough: Catherine Romatowski, who had lost the ability to whisper due to severe ME, joined the rapamycin trial and regained her voice after years of silence, though she noted that recovery remains a slow and non-linear process.
- The Non-Responders: Not everyone sees a benefit. Some users reported “feeling sick as a dog” for a week after their first dose, while others discontinued treatment due to painful mouth sores (stomatitis) or lack of perceived benefit within the first 30 days.
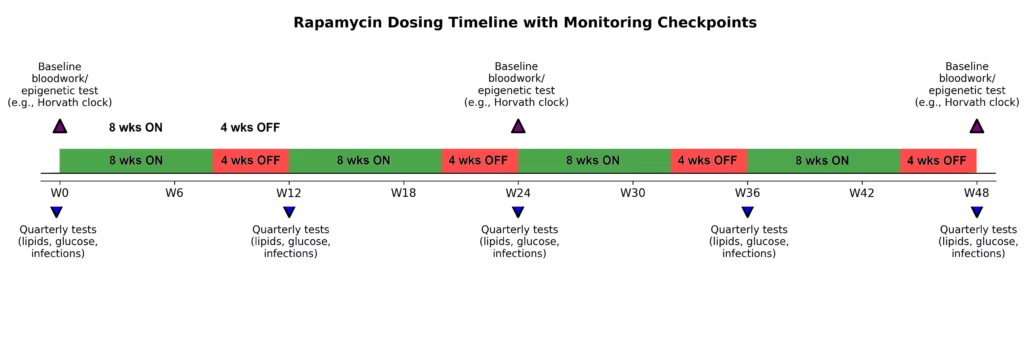
What are the risks and standard dosing protocols for 2026?
Low-dose rapamycin used for Long COVID (typically 1–6 mg once per week) has a significantly safer profile than the high-dose daily regimens used in organ transplants. At these “pulsatile” doses, the drug acts as an immune enhancer rather than a suppressor.
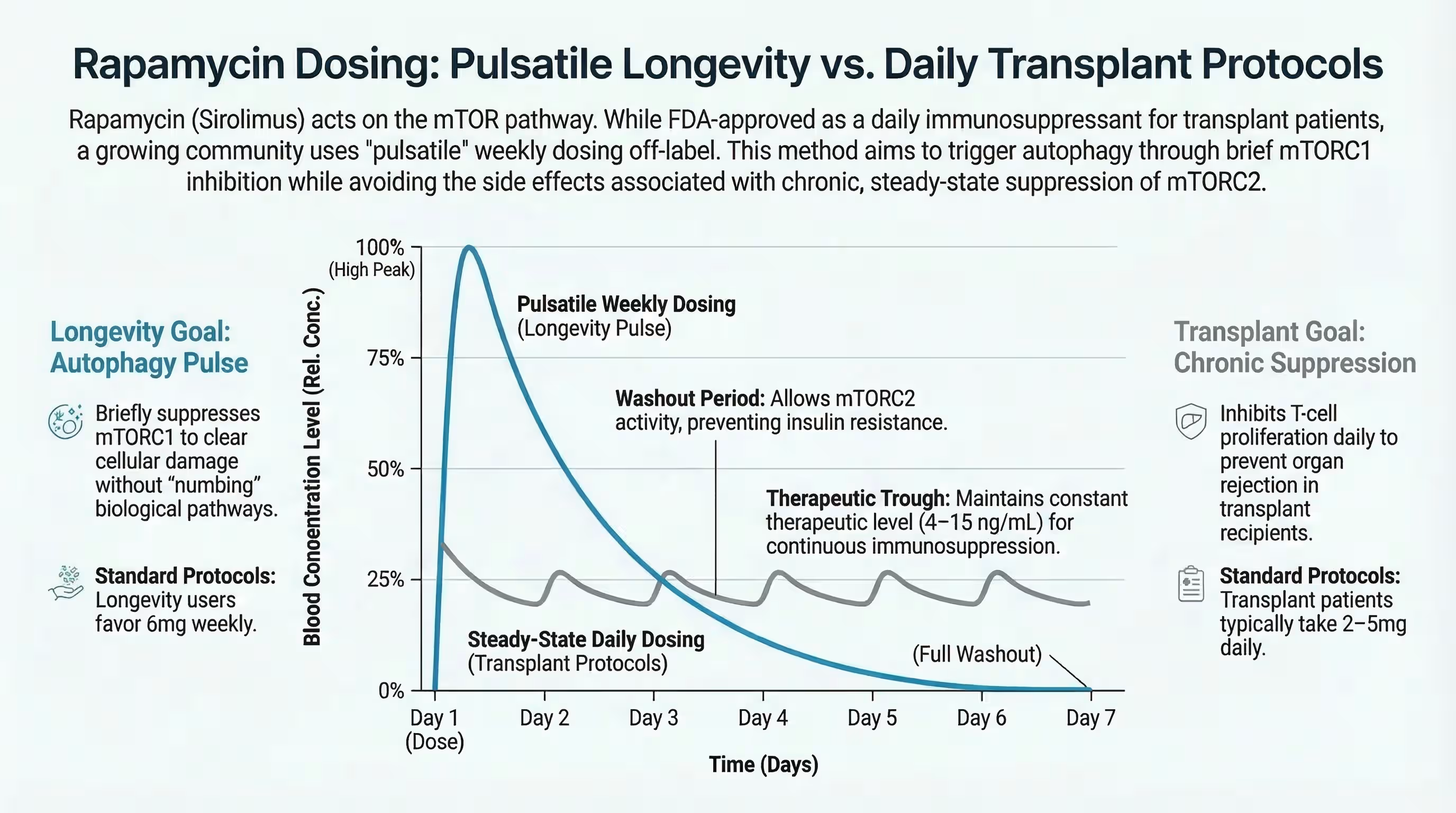
Standard “Longevity Dose” Protocol:
- Titration: Most clinicians start patients at 1mg per week and gradually increase by 1mg each week until reaching a target dose of 4–6mg.
- Safety Monitoring: Monthly labs are essential to track lipids (cholesterol) and glucose levels, as rapamycin can occasionally cause mild elevations.
- Cycling: Many experts, including Dr. Kaufman, recommend taking regular breaks from the drug to prevent blocking the mTORC2 complex, which is vital for long-term health.
Common Side Effects:
- Headaches (often temporary)
- Mouth sores (canker sores/stomatitis)
- Elevated triglycerides or blood glucose
- Transient insomnia

Conclusion: The Future of Rapamycin in 2026
The trend for 2026 is clear: we are moving away from “fixing aging” and toward “biological coordination”. Rapamycin has proven itself to be a powerful tool for restoring that coordination in people whose lives have been hijacked by Long COVID or ME/CFS. While it is not a “magic pill” for every patient, the ability to identify responders through biomarkers like pATG13 marks a new era of precision medicine for complex chronic illnesses.
Weigh benefits against monitored risks—it’s not a cure-all.
Ready to explore? Rapashop offers lab-tested rapamycin with global shipping. The 2026 consensus is to work with a clinician who understands low-dose titration and the necessity of monitoring your metabolic markers.

No, it’s off-label; trials like Mount Sinai’s probe this.
It is not an “on/off switch.” Data suggests that while some feel better immediately, clinical improvements in autophagy markers and fatigue often peak between 3 to 6 months of consistent weekly dosing.
Most patients report improved mood and reduced brain fog. However, some clinical reviews note that mTOR inhibitors can occasionally be associated with increased anxiety or noradrenaline in specific populations.
No—screen for contraindications via MD.
Yes, via mitochondrial support in trials.
2mg weekly, titrate slowly.
Resource links
Clinical trials are testing cancer drug rapamycin for Long COVID and ME
Rapamycin Pilot Treatment Trial for ME/CFS
YOUTUBE: How Rapamycin Supports Longevity Through Autophagy
Simmaron’s Solve ME/CFS Catalyst Award-Winning Rapamycin Trial Yields Promising Treatment Findings
YOUTUBE: David Putrino | International ME/CFS Conference 2025: Low-dose Rapamycin for Long COVID
Long COVID low-dose rapamycin clinical trial
Simmaron’s Rapamycin ME/CFS Trial Moves Forward: The Goal – FDA Approval