Animal Studies on Rapamycin: From Mice to Men – Can We Translate the 60% Lifespan Extension?

The translation of mammalian target of rapamycin (mTOR) inhibitors from preclinical models to human longevity interventions is the most scrutinized frontier in modern geroscience. We are moving far beyond the theoretical question of if rapamycin extends lifespan. Decades of data confirm its efficacy across yeast, nematodes, fruit flies, and mice. Today, the clinical challenge is translation: How do we safely harness intermittent mTORC1 inhibition to delay age-related diseases in humans without triggering the toxicities seen in transplant patients?
With the recent conclusion of the PEARL (Participatory Evaluation of Aging with Rapamycin for Longevity) trial, the launch of the Test of Rapamycin in Aging Dogs (TRIAD) study, and novel applications for post-viral syndromes, researchers are finally decoding the nuanced leap from murine models to human clinical application.
Here is a deep dive into the latest clinical data, the critical distinction between daily and pulsatile dosing, and the reality of translating animal longevity breakthroughs into human healthspan extension.
How Much Does Rapamycin Extend Life in Animal Studies?
In murine models, continuous rapamycin administration extends median lifespan by 9% to 14%, while transient, high-dose treatments in middle-aged mice have increased remaining life expectancy by up to 60%. These robust findings establish rapamycin as the most consistent pharmacological agent for lifespan extension in mammals.
The exact degree of lifespan extension depends heavily on the intervention’s timing, dosage, and the subject’s sex. In a landmark study, researchers tested a transient three-month treatment of rapamycin (8 mg/kg/day via injection) on middle-aged mice—roughly equivalent to 60-65 human years. Following this short-term intervention, the male mice experienced a staggering 60% increase in post-treatment life expectancy.
However, translating these results requires extreme caution regarding dose and sex-specific responses. In that same high-dose injection study, female mice did not experience any lifespan extension. Instead, the profound mTOR suppression triggered a striking shift toward aggressive hematopoietic cancers (like histiocytic sarcoma and lymphoma), which completely neutralized the survival benefit.
Conversely, when researchers utilized a lower, dietary dose (126 parts per million) over the same three-month window, both male and female mice saw substantial life expectancy increases—up to 42% pooled—without the aggressive oncological side effects. Furthermore, the treated mice exhibited persistent improvements in motor coordination and grip strength long after the drug was withdrawn. This highlights a critical principle for human translation: the geroprotective benefits of rapamycin are tightly bound to a precise therapeutic window where “more” does not equal “better.”

How Does Rapamycin Inhibit the mTORC1 Pathway?
Rapamycin acts as an allosteric inhibitor that selectively suppresses the mTORC1 protein complex, shifting cellular machinery away from continuous protein synthesis and into a repair state. This targeted suppression triggers macroautophagy, clearing out senescent cells and misfolded proteins that drive biological aging.
To understand exactly what does rapamycin inhibit, we must differentiate between two distinct protein complexes: mTORC1 and mTORC2.
- mTORC1 acts as the body’s primary nutrient sensor and biological “accelerator pedal.” It promotes cellular growth, proliferation, and anabolic metabolism. Aging essentially traps this pedal in the “on” position, leading to cellular hyperfunction, the accumulation of toxic molecular debris, and the exhaustion of stem cells.
- mTORC2 regulates foundational metabolic processes, including insulin sensitivity, cellular survival, and cytoskeletal organization.
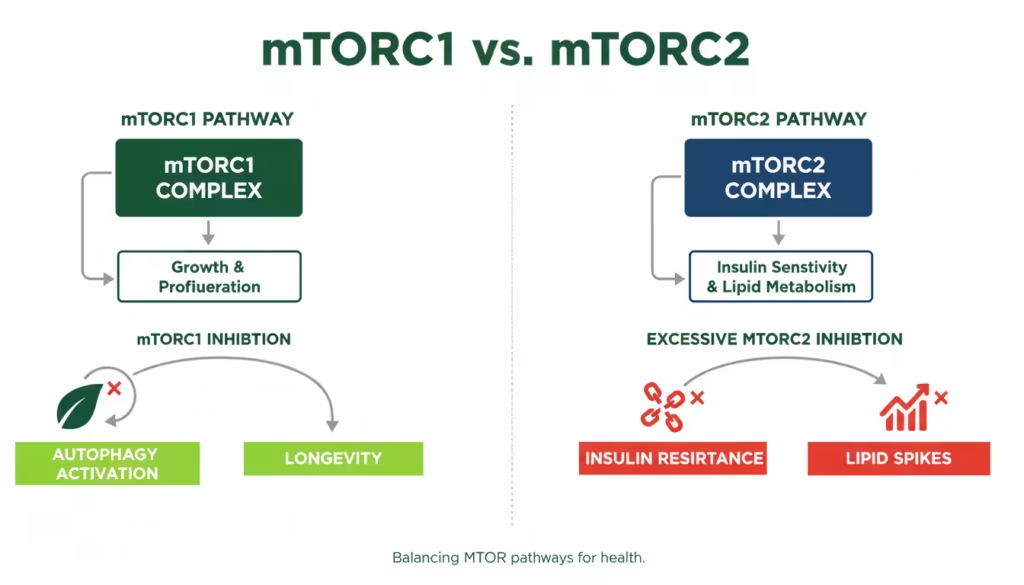
The clinical goal for longevity is selective inhibition. Intermittent rapamycin administration reliably suppresses mTORC1 to trigger autophagy without severely blunting mTORC2. If mTORC2 is chronically suppressed—as seen in daily transplant-level dosing—patients rapidly develop insulin resistance, hyperlipidemia, and clinical immunosuppression.
What Did the PEARL Trial Reveal About Human Translation?
The PEARL trial demonstrated that low-dose, weekly rapamycin (5–10 mg) is safe for healthy older adults over a 48-week period, yielding modest gains in lean muscle mass and reductions in subjective pain. Importantly, the trial highlighted that compounded rapamycin suffers from significantly lower bioavailability compared to generic formulations.
The Participatory Evaluation of Aging with Rapamycin for Longevity (PEARL) trial represents a massive leap in human geroscience. Utilizing a decentralized, double-blind, randomized, placebo-controlled design, researchers tested rapamycin in normative-aging adults.
While the study did not show dramatic drops in visceral fat (the primary endpoint), the secondary body composition metrics were highly encouraging. Women taking the 10 mg weekly dose experienced a statistically significant 4.5% increase in lean tissue mass, alongside notable improvements in self-reported pain scores. Male participants in the 10 mg cohort saw trending increases in bone mineral content.
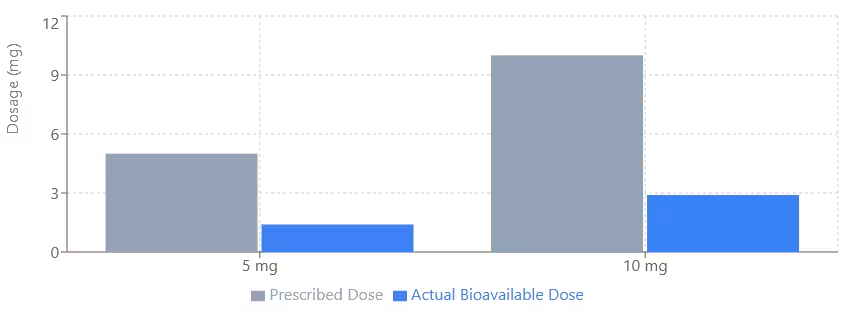
Crucially, the PEARL trial illuminated a massive variable in off-label rapamycin protocols: formulation and absorption. Post-trial pharmacokinetic analysis revealed that the compounded rapamycin used in the study was roughly 3.5 times less bioavailable than commercially manufactured generic sirolimus. This means the 10 mg cohort was functionally receiving an exposure closer to 2.9 mg of generic rapamycin. The fact that physiological benefits were observed at such a low functional dose makes the clinical case for low-dose mTOR inhibition highly compelling.
What is the Standard Longevity Dose for Rapamycin in 2026
The emerging standard longevity dose for rapamycin relies on a pulsatile schedule of 3 to 10 mg taken once weekly. This intermittent protocol ensures acute mTORC1 suppression to stimulate autophagy, followed by a drug-clearance window that prevents chronic mTORC2 inhibition.
Steps for Evaluating Pulsatile vs. Daily Dosing Protocols:
- Calculate Relative Exposure: Leading clinical researchers suggest a relative target range of 0.075 to 0.15 mg/kg once weekly. For a 70 kg adult, this scales to roughly 5 to 10 mg per week.
- Verify Formulation: Because compounded pharmacy products have shown significantly lower bioavailability in recent trials, clinicians generally recommend standard generic sirolimus tablets.
- Implement Dosing Vacations: To maintain physiological flexibility, many physicians suggest cycling the drug (e.g., 8–12 weeks on, followed by 4 weeks off) to ensure immune and metabolic systems fully reset.
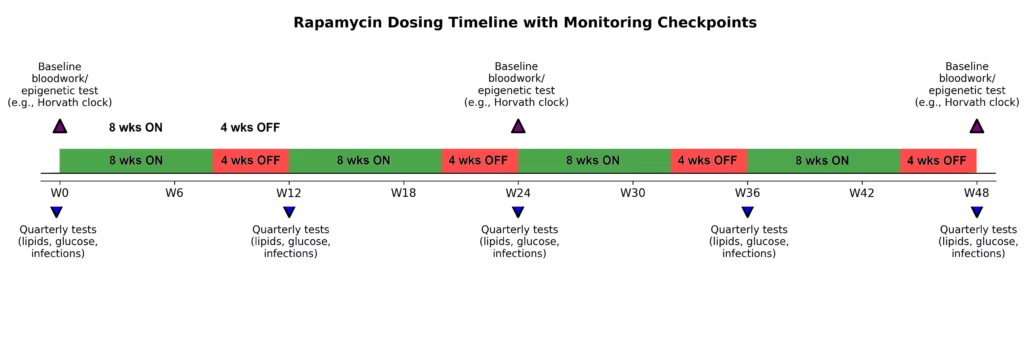
Expert consensus remains cautiously optimistic but grounded in data. Dr. Peter Attia categorizes rapamycin as one of the most “promising” geroprotective molecules available, specifically because its mechanism elegantly mimics the longevity benefits of caloric restriction. Unlike fasting, which provides an indirect and variable suppression of mTOR, rapamycin acts as a direct pharmacologic inhibitor. However, Dr. Attia and leading geroscientists like Dr. Matt Kaeberlein continually stress that off-label longevity use remains experimental and requires strict medical supervision, as human lifespan data is not yet definitive.
Can I Get Rapamycin for My Dog?
Yes, rapamycin is increasingly being prescribed off-label by veterinarians to promote healthspan and treat age-related decline in dogs. The optimal dog rapamycin dose, currently under rigorous investigation in the Dog Aging Project, is typically 0.15 mg/kg administered once weekly.
Canine models are the perfect translational bridge between mice and humans because companion dogs share our environments, disease profiles, and aging trajectories.
The Test of Rapamycin in Aging Dogs (TRIAD) trial is actively tracking the safety and efficacy of weekly rapamycin (0.15 mg/kg) in a massive, double-masked, placebo-controlled format. Earlier 10-week pilot studies already demonstrated that low-dose rapamycin improves age-related cardiac function—specifically left ventricular fractional shortening—with a highly favorable safety profile.
Many pet owners involved in early cohorts or utilizing the drug off-label report rapid returns of energy and delayed progression of canine cognitive dysfunction. Because dogs age seven to ten times faster than humans, the TRIAD trial will provide definitive answers on mammalian lifespan extension long before human trials conclude.

Is Rapamycin Effective for Long Covid or ME/CFS Symptoms?
Emerging clinical data suggests low-dose rapamycin significantly alleviates fatigue and post-exertional malaise (PEM) in ME/CFS and Long COVID patients. The drug appears to restore impaired autophagy, as indicated by elevated Beclin-1 levels and reduced mTOR overactivation in pilot studies.
One of the most exciting developments in 2025–2026 is the repurposing of rapamycin for infection-associated chronic illnesses (IACCIs). Two major clinical trials—one at Mount Sinai’s Cohen Center for Recovery from Complex Chronic Illness (CoRE) and another by Simmaron Research—are evaluating low-dose rapamycin (titrated up to 6 mg/week) for Long COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS).
In ME/CFS, the mTOR pathway is frequently overactivated, which blocks autophagy. This means cells cannot renew energy-producing mitochondria fast enough, driving severe muscle fatigue and PEM. Early preprint data from the Simmaron trial shows that 72.5% of patients taking 6 mg/week experienced notable improvements in fatigue, PEM, and orthostatic intolerance. Biomarker analysis confirmed this clinical relief aligned with restored autophagy: patients showed increased plasma Beclin-1 (an autophagy marker) and suppressed pSer258-ATG13 (a marker of mTOR overactivation).
Real User Experience Beyond the clinical data, real-world patient sentiment highlights both the miraculous potential and the stark reality of biological variability. In chronic illness forums like r/cfs, users tracking their off-label use note profound shifts. A 26-year-old Long COVID patient named Alex reported going from “bed bound with severe post-exertional malaise to playing basketball in six weeks” after starting 5mg of weekly rapamycin via a telehealth clinic.
However, the clinical reality is nuanced. Six months later, Alex experienced a relapse, illustrating that while rapamycin clears biological waste, it is not always a permanent cure. Another patient, Wade, reported feeling “weightless” and having a miraculous return of energy, but had to discontinue the drug by week three due to severe mouth sores and allergic reactions. This underscores the necessity of personalized, closely monitored dosing.
What Are the Potential Side Effects of Pulsatile Rapamycin?
The most common side effects of intermittent rapamycin include mild mouth ulcers (aphthous stomatitis), transient gastrointestinal upset, and hyperlipidemia. By adhering to a weekly dosing schedule rather than daily administration, users largely bypass severe risks like clinical immunosuppression or insulin resistance.
Understanding the side effect profile requires separating daily transplant dosing from weekly longevity dosing.
| Side Effect | Daily Dosing (Transplant / Oncology) | Weekly Dosing (Longevity / Off-label) | Mitigation Strategies |
|---|---|---|---|
| Mouth Ulcers | 44–66% incidence | 5–15% incidence | Topical dexamethasone rinse, B12 lozenges. |
| Hyperlipidemia | High incidence; marked LDL/Triglyceride spikes | Mild to moderate transient increases | Monitor every 3 months; omega-3s, dose adjustment. |
| Immunosuppression | Intentional clinical suppression (T-cell block) | Paradoxical immune enhancement | Withhold dose during active bacterial/viral infections. |
| Insulin Resistance | High risk due to chronic mTORC2 inhibition | Low risk; neutral or improved sensitivity | Routine HbA1c and fasting glucose monitoring. |
TABLE: Pulsatile vs. Daily Dosing of Rapamycin: Side Effect Profiles
Does low-dose rapamycin cause mouth sores? Yes, aphthous stomatitis remains the most frequent complaint even at 5 mg a week, usually appearing within the first month of treatment.
Will rapamycin make me immunocompromised if I take it weekly? This is a common misconception. While 5 mg daily suppresses the immune system to prevent organ rejection, 5 mg weekly actually boosts immune function in older adults. A landmark 2014 study demonstrated that a six-week course of low-dose mTOR inhibitors enhanced the immune response to the influenza vaccine by 20% in elderly volunteers.

Summary of Key Takeaways
Profound Animal Efficacy: Rapamycin consistently extends lifespan by 9-14% across mammalian models, with transient high doses pushing post-treatment life expectancy up to 60% in male cohorts.
Targeted Autophagy: By selectively inhibiting the mTORC1 pathway, rapamycin triggers macroautophagy and mitochondrial recycling without chronically suppressing the metabolically crucial mTORC2 pathway.
Human Safety Profile Established: The PEARL trial confirmed that a longevity dose of 5–10 mg weekly is safe, yielding positive shifts in lean muscle mass and joint pain.
Emerging ME/CFS Therapeutics: 6mg weekly doses are actively restoring autophagy and alleviating severe fatigue in Long COVID and ME/CFS clinical trials.
Individualized Risk: Side effects like mouth ulcers and elevated lipids are strictly dose-dependent. A “start low, go slow” pulsatile protocol is imperative.
Before incorporating geroprotective compounds into your routine, consult with a longevity-focused physician to establish baseline blood markers (CBC, lipid panel, HbA1c).

No direct evidence; mouse 60% gains don’t scale due to biology differences. Trials show healthspan perks.
3-10mg weekly, intermittent; monitor bloodwork.
Rare; mostly mild GI or sores, manageable.
Early ME/CFS data: 72.5% improved.
No, off-label only.
While some longevity clinics prescribe this “stack,” Dr. Peter Attia and others note that the geroprotective evidence for metformin in non-diabetics is becoming “fuzzy.” Both drugs manipulate cellular energy sensing (mTOR and AMPK), but combining them should be done under strict medical supervision to avoid compounded gastrointestinal distress or blood glucose fluctuations.
No. While daily dosing is used to intentionally suppress the immune system for organ transplants, weekly low-dose regimens have actually been shown to enhance immune function. Clinical trials report a ~20% increase in immune response titers to influenza vaccines in older adults taking weekly rapalogs.
Yes, aphthous stomatitis (mouth ulcers) is the most common side effect, even at low longevity doses. They usually occur within the first few weeks of treatment and can be mitigated with topical steroid rinses or by slightly lowering the weekly dose.
While intermittent fasting naturally lowers mTOR and boosts AMPK to stimulate autophagy, rapamycin provides a direct, pharmacologic inhibition of mTORC1. Fasting effects are highly variable based on diet and tissue type, whereas rapamycin offers a consistent, titratable response without the muscle-loss risks associated with prolonged caloric restriction.
The current consensus for off-label longevity use is between 3 mg and 10 mg taken once weekly. This allows the drug to peak and trough, inhibiting mTORC1 for autophagy while clearing the system fast enough to avoid suppressing mTORC2.
Daily, high-dose rapamycin suppresses mTORC2, which can lead to insulin resistance and elevated blood glucose. However, weekly low-dose regimens typically avoid this complication, and routine HbA1c monitoring is used to ensure metabolic stability.
Resource links
Transient rapamycin treatment can increase lifespan and healthspan in middle-aged mice
Rapamycin as a potential intervention to promote longevity and extend healthspan in companion dogs
First Results from the PEARL Trial of Rapamycin
Influence of rapamycin on safety and healthspan metrics after one year: PEARL trial results
Rapamycin: A drug that may slow aging with Matt Kaeberlein, PhD
Test of Rapamycin in Aging Dogs (TRIAD)
Clinical trials are testing cancer drug rapamycin for Long COVID and ME
Targeting Autophagy: A Clinical Trial of Low-Dose Rapamycin for ME/CFS, Long COVID, and IACCIs
What are the protocols for using rapamycin (sirolimus) for longevity?




